Comparison of gemcitabine plus oxaliplatin versus gemcitabine plus nab-paclitaxel as first-line chemotherapy for advanced pancreatic adenocarcinoma: A single-center retrospective analysis
- PMID: 37537780
- PMCID: PMC10501299
- DOI: 10.1002/cam4.6334
Comparison of gemcitabine plus oxaliplatin versus gemcitabine plus nab-paclitaxel as first-line chemotherapy for advanced pancreatic adenocarcinoma: A single-center retrospective analysis
Abstract
Background: Pancreatic cancer is mostly diagnosed in an advanced stage and treated with systemic therapy with palliative intent. Nowadays, the doublet chemotherapy of Gemcitabine and nab-paclitaxel (Gem-Nab) is one of the most frequently used regimens worldwide, but is not ubiquitarily available or reimbursed. Therefore, we compared the clinical efficacy of Gem-Nab to a historical control of patients treated with gemcitabine and oxaliplatin (Gem-Ox) at our tertiary cancer center, which was the standard treatment prior to the introduction of FOLFIRINOX.
Methods: This single-center retrospective real world study includes 121 patients diagnosed with locally advanced or primary metastatic pancreatic adenocarcinoma who were treated with chemotherapy doublet, with either Gem-Nab or Gem-Ox in palliative first-line. Survival rates were analyzed using the Kaplan-Meier method, and comparisons were made with log-rank tests. Gem-Ox was considered as standard first line therapy at our institution for patients who were deemed fit for doublet chemotherapy between the years 2006 to 2012. These patients were compared to a cohort of patients treated with the new standard first-line therapy of Gem-Nab between 2013 and 2020.
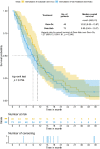
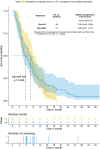
Results: A total of 554 patients with pancreatic cancer of all stages were screened, and 73 patients treated with Gem-Nab and 48 patients treated with Gem-Ox in the palliative first-line setting were identified and included in this analysis. Patients receiving Gem-Ox had a statistically significantly better performance score (ECOG PS) when compared to the Gem-Nab group (Odds ratio (OR) 0.28, 95% CI 0.12-0.65, p = 0.005), more often suffered from locally advanced than metastatic disease (OR 3.10, 95% CI 1.27-7.91, p = 0.019) and were younger in age (OR 0.95, 95% CI 0.91-0.99, p = 0.013). Median overall survival (OS) of the whole study cohort was 10.3 months (95% CI 8.5-11.6). No statistically significant difference in OS could be observed between the Gem-Nab and the Gem-Ox cohort (median OS: 8.9 months (95% CI 6.4-13.5) versus 10.9 months (95% CI 9.5-13.87, p = 0.794, HR 1.27, 95% CI 0.85-1.91)). Median progression-free survival (PFS) was 6.8 months in the entire cohort (95% CI 4.9-8.4). No statistically significant difference in PFS could be observed between the Gem-Nab and the Gem-Ox cohort (median PFS: 5.8 months (95% CI 4.3-8.2) versus 7.9 months (95% CI 5.4-9.5) p = 0.536, HR 1.11, 95% CI 0.74-1.67). Zero-truncated negative binomial regressions on OS and PFS adjusting for gender, age, performance status (ECOG PS), and CA19-9 levels yielded no significant difference between Gem-Nab or Gem-Ox.
Conclusion: From our analysis, we could evidence no difference in outcome parameters in this retrospective analysis despite the worse prognostic pattern for GemOX. Therefore, we suggest Gem-Ox as potential first line treatment option for inoperable locally advanced or metastatic pancreatic cancer, especially if Gem-Nab is not available.
Keywords: GEMOX; gemcitabine; nab-paclitaxel; oxaliplatin; pancreatic cancer; treatment costs.
© 2023 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
References
-
- Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913‐2921. - PubMed
-
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209‐249. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials