Initiation and duration of folic acid supplementation in preventing congenital malformations
- PMID: 37545008
- PMCID: PMC10405478
- DOI: 10.1186/s12916-023-03000-8
Initiation and duration of folic acid supplementation in preventing congenital malformations
Abstract
Background: Folic acid (FA) supplementation is associated with a lower risk of the neural tube and heart defects and is recommended for women of childbearing age. Although there are detailed recommendations, differences in the initiation time and duration of FA supplementation remain poorly studied.
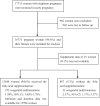
Methods: A multicentre prospective study of 17,713 women was conducted. The incidence of congenital malformations in women taking a recommended dosage (e.g. 0.4 or 0.8 mg/day) of FA was compared with that in women without supplementation. The predicted probability of malformations by the initiation time and duration of FA use was estimated to determine optimal options.
Results: Periconceptional FA supplementation was associated with a lower and insignificant risk of congenital malformations (1.59% vs. 2.37%; odds ratio [OR] 0.69; 95% confidence interval [CI]: 0.44-1.08), heart defects (3.8 vs. 8.0 per 1000 infants; OR, 0.47; 0.21-1.02), and neural tube defects (7.0 vs. 11.5 per 10,000 infants; OR, 0.64; 0.08-5.15). FA use after pregnancy provided greater protection against total malformations. Statistically significant associations were found in women who initiated FA supplementation in the first month of gestation (OR, 0.55; 95% CI: 0.33-0.91) and in those who supplemented for 1 to 2 months (OR, 0.59; 95% CI: 0.36-0.98). Similar results were found for heart defects. The optimal initiation time was 1.5 (optimal range: 1.1 to 1.9) months before pregnancy and a duration of 4.0 (3.7 to 4.4) months was reasonable to achieve the lowest risk of congenital malformations. Heart defect prevention required an earlier initiation (2.2 vs. 1.1 months before pregnancy) and a longer duration (4.7 vs. 3.7 months) than the prevention of other malformations.
Conclusions: The timely initiation of FA supplementation for gestation was associated with a decreased risk of congenital malformations, which was mainly attributed to its protection against heart defects. The initiation of FA supplementation 1.5 months before conception with a duration of 4 months is the preferred option for congenital malformation prevention.
Trial registration: Chictr.org.cn identifier: ChiCTR-SOC-17010976.
Keywords: Cohort study; Congenital malformation; Folic acid supplementation; Heart defects; Neural tube defects.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Sarmah S, Muralidharan P, Marrs JA. Common congenital anomalies: environmental causes and prevention with folic acid containing multivitamins. Birth Defects Res C Embryo Today. 2016;108:274–286. - PubMed
-
- Christianson AL, Howson CP, Modell B. March of dimes global report on birth defects. The hidden toll of dying and disabled children. White Plains, New York. 2006. http://www.marchofdimes.org/materials/global-report-on-birth-defectsthe-...
-
- Viswanathan M, Treiman KA, Kish-Doto J, Middleton JC, Coker-Schwimmer EJ, Nicholson WK. Folic acid supplementation for the prevention of neural tube defects: an updated evidence report and systematic review for the US Preventive Services Tak Force. JAMA. 2017;317(2):190–203. - PubMed
-
- Czeizel AE. Periconceptional folic acid and multivitamin supplementation for the prevention of neural tube defects and other congenital abnormalities. Birth Defects Res A Clin Mol Teratol. 2009;85:260–268. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

