Proteolipid Protein-Induced Mouse Model of Multiple Sclerosis Requires B Cell-Mediated Antigen Presentation
- PMID: 37548478
- PMCID: PMC10528642
- DOI: 10.4049/jimmunol.2200721
Proteolipid Protein-Induced Mouse Model of Multiple Sclerosis Requires B Cell-Mediated Antigen Presentation
Abstract
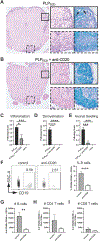
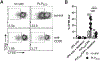
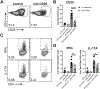
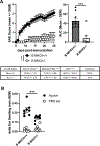
The pathogenic role B cells play in multiple sclerosis is underscored by the success of B cell depletion therapies. Yet, it remains unclear how B cells contribute to disease, although it is increasingly accepted that mechanisms beyond Ab production are involved. Better understanding of pathogenic interactions between B cells and autoreactive CD4 T cells will be critical for novel therapeutics. To focus the investigation on B cell:CD4 T cell interactions in vivo and in vitro, we previously developed a B cell-dependent, Ab-independent experimental autoimmune encephalomyelitis (EAE) mouse model driven by a peptide encompassing the extracellular domains of myelin proteolipid protein (PLPECD). In this study, we demonstrate that B cell depletion significantly inhibited PLPECD-induced EAE disease, blunted PLPECD-elicited delayed-type hypersensitivity reactions in vivo, and reduced CD4 T cell activation, proliferation, and proinflammatory cytokine production. Further, PLPECD-reactive CD4 T cells sourced from B cell-depleted donor mice failed to transfer EAE to naive recipients. Importantly, we identified B cell-mediated Ag presentation as the critical mechanism explaining B cell dependence in PLPECD-induced EAE, where bone marrow chimeric mice harboring a B cell-restricted MHC class II deficiency failed to develop EAE. B cells were ultimately observed to restimulate significantly higher Ag-specific proliferation from PLP178-191-reactive CD4 T cells compared with dendritic cells when provided PLPECD peptide in head-to-head cultures. We therefore conclude that PLPECD-induced EAE features a required pathogenic B cell-mediated Ag presentation function, providing for investigable B cell:CD4 T cell interactions in the context of autoimmune demyelinating disease.
Copyright © 2023 by The American Association of Immunologists, Inc.
Figures

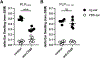




References
-
- Culpepper WJ, Marrie RA, Langer-Gould A, Wallin MT, Campbell JD, Nelson LM, Kaye WE, Wagner L, Tremlett H, Chen LH, et al. (2019). Validation of an algorithm for identifying MS cases in administrative health claims datasets. Neurology 92, e1016–e1028. 10.1212/WNL.0000000000007043. - DOI - PMC - PubMed
-
- Wallin MT, Culpepper WJ, Campbell JD, Nelson LM, Langer-Gould A, Marrie RA, Cutter GR, Kaye WE, Wagner L, Tremlett H, et al. (2019). The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology 92, e1029–e1040. 10.1212/WNL.0000000000007035. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

