Ca2+ signaling and the Hippo pathway: Intersections in cellular regulation
- PMID: 37549859
- PMCID: PMC10529277
- DOI: 10.1016/j.cellsig.2023.110846
Ca2+ signaling and the Hippo pathway: Intersections in cellular regulation
Abstract
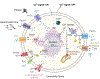
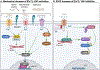
The Hippo signaling pathway is a master regulator of organ size and tissue homeostasis. Hippo integrates a broad range of cellular signals to regulate numerous processes, such as cell proliferation, differentiation, migration and mechanosensation. Ca2+ is a fundamental second messenger that modulates signaling cascades involved in diverse cellular functions, some of which are also regulated by the Hippo pathway. Studies published over the last five years indicate that Ca2+ can influence core Hippo pathway components. Nevertheless, comprehensive understanding of the crosstalk between Ca2+ signaling and the Hippo pathway, and possible mechanisms through which Ca2+ regulates Hippo, remain to be elucidated. In this review, we summarize the multiple intersections between Ca2+ and the Hippo pathway and address the biological consequences.
Keywords: Calcium; Calmodulin; Hippo; S100; Signaling; YAP.
Published by Elsevier Inc.
Conflict of interest statement
Declaration of Competing Interest Authors declare no competing interest.
Figures



References
-
- Hilman D, Gat U, The evolutionary history of YAP and the hippo/YAP pathway, Mol Biol Evol 28(8) (2011) 2403–17. - PubMed
-
- Berridge MJ, Lipp P, Bootman MD, The versatility and universality of calcium signalling, Nat Rev Mol Cell Biol 1(1) (2000) 11–21. - PubMed
-
- Ma S, Meng Z, Chen R, Guan KL, The Hippo Pathway: Biology and Pathophysiology, Annu Rev Biochem 88 (2019) 577–604. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

