Comparative Safety Surveillance of Triple (IDA) Versus Dual Therapy (DA) in Mass Drug Administration for Elimination of Lymphatic Filariasis in Kenya: A Cohort Event Monitoring Study
- PMID: 37552438
- PMCID: PMC10584738
- DOI: 10.1007/s40264-023-01338-9
Comparative Safety Surveillance of Triple (IDA) Versus Dual Therapy (DA) in Mass Drug Administration for Elimination of Lymphatic Filariasis in Kenya: A Cohort Event Monitoring Study
Abstract
Introduction: Dual diethylcarbamazine and albendazole (DA) therapy is the standard mass drug administration (MDA) regimen for lymphatic filariasis in Kenya. Following the recent World Health Organization recommendation, Kenya piloted triple therapy with ivermectin, diethylcarbamazine, and albendazole (IDA) in MDA.
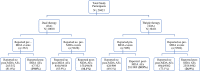
Objective: We conducted a community-based, observational, cohort event monitoring study to compare the types, frequency, severity, and predictors of adverse events following dual versus triple therapy in 20,421 eligible residents.
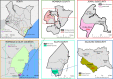
Methods: Residents in Kilifi (n = 10,010) and Mombasa counties (n = 10,411) received DA and IDA through MDA campaigns, respectively. Adverse events were actively monitored through house-to-house visits on days 1, 2, and 7 after MDA. Any clinical events reported before and after MDA were cross-checked and verified to differentiate pre-existing events from MDA-associated adverse events.
Results: Overall, 5807 and 3102 adverse events were reported by 2839 and 1621 individuals in the IDA and DA groups, respectively. The incidence of experiencing one or more adverse events was significantly higher (p < 0.0001) in the IDA group (27.3%; 95% confidence interval [CI] 26.4-28.2) than in the DA group (16.2%; 95% CI 15.5-16.9). Dizziness (15.9% vs 5.9%) and drowsiness (10.1% vs 2.6%) were the most common adverse events and significantly higher in the IDA group compared with the DA group (p < 0.0001). Most adverse events were mild or moderate with a few severe cases (IDA = 0.05%; 95% CI 0.35-0.78, DA = 0.03%; 95% CI 0.14-0.60). Female sex, obesity, taking three or more diethylcarbamazine or ivermectin tablets, and having pre-existing clinical symptoms were significant predictors of adverse events following IDA treatment.
Conclusions: Ivermectin, diethylcarbamazine, and albendazole as a combination is as safe and well tolerated as DA to use in MDA campaigns with no serious life-threatening adverse events. Systemic mild-to-moderate adverse events with a few severe cases and transient adverse events are more common with IDA treatment than with DA treatment. Hence, integrating pharmacovigilance into a MDA program is recommended for the timely detection and management of adverse events.
© 2023. The Author(s).
Conflict of interest statement
Christabel Khaemba, Abbie Barry, Wyckliff P. Omondi, Elvis Kirui, Margaret Oluka, Gurumurthy Parthasarathi, Sammy M. Njenga, Anastacia Guantai, and Eleni Aklillu have no conflicts of interest that are directly relevant to the content of the article.
Figures

References
-
- World Health Organization. Global programme to eliminate lymphatic filariasis: progress report, 2017. 2018. https://apps.who.int/iris/bitstream/handle/10665/275719/WER9344.pdf?ua=1. Accessed 20 Feb 2023.
-
- World Health Organization. Weekly epidemiological record record No. 43, 23 October 2020. 2020. https://apps.who.int/iris/bitstream/handle/10665/336185/WER9543-eng-fre.pdf. Accessed 26 Feb 2023.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

