Ssa1-targeted antibody prevents host invasion by Candida albicans
- PMID: 37560525
- PMCID: PMC10407798
- DOI: 10.3389/fmicb.2023.1182914
Ssa1-targeted antibody prevents host invasion by Candida albicans
Abstract
Introduction: Candida albicans is a commensal fungus that colonizes most healthy individuals' skin and mucosal surfaces but can also cause life-threatening invasive infections, particularly in immunocompromised patients. Despite antifungal treatment availability, drug resistance is increasing, and mortality rates remain unacceptably high. Heat shock protein Ssa1, a conserved member of the Hsp70 family in yeast, is a novel invasin that binds to host cell cadherins, induces host cell endocytosis, and enables C. albicans to cause maximal damage to host cells and induces disseminated and oropharyngeal disease.
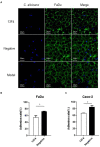
Result: Here we discovered a mouse monoclonal antibody (mAb 13F4) that targeting C. albicans Ssa1 with high affinity (EC50 = 39.78 ng/mL). mAb 13F4 prevented C. albicans from adhering to and invading human epithelial cells, displayed antifungal activity, and synergized with fluconazole in proof of concept in vivo studies. mAb 13F4 significantly prolonged the survival rate of the hematogenous disseminated candidiasis mice to 75%. We constructed a mAb 13F4 three-dimensional structure using homology modeling methods and found that the antigen-binding fragment (Fab) interacts with the Ssa1 N-terminus.
Discussion: These results suggest that blocking Ssa1 cell surface function may effectively control invasive C. albicans infections and provide a potential new treatment strategy for invasive fungal infections.
Keywords: Candida albicans; Ssa1; antibody; antifungal; systemic infection.
Copyright © 2023 Qiu, Shen, Jiang, Ji, Zhang, Hou, Zhang, Shen and An.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
LinkOut - more resources
Full Text Sources

