Tetraspanins set the stage for bone marrow microenvironment-induced chemoprotection in hematologic malignancies
- PMID: 37561544
- PMCID: PMC10432613
- DOI: 10.1182/bloodadvances.2023010476
Tetraspanins set the stage for bone marrow microenvironment-induced chemoprotection in hematologic malignancies
Abstract
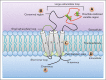
Despite recent advances in the treatment of hematologic malignancies, relapse still remains a consistent issue. One of the primary contributors to relapse is the bone marrow microenvironment providing a sanctuary to malignant cells. These cells interact with bone marrow components such as osteoblasts and stromal cells, extracellular matrix proteins, and soluble factors. These interactions, mediated by the cell surface proteins like cellular adhesion molecules (CAMs), induce intracellular signaling that leads to the development of bone marrow microenvironment-induced chemoprotection (BMC). Although extensive study has gone into these CAMs, including the development of targeted therapies, very little focus in hematologic malignancies has been put on a family of cell surface proteins that are just as important for mediating bone marrow interactions: the transmembrane 4 superfamily (tetraspanins; TSPANs). TSPANs are known to be important mediators of microenvironmental interactions and metastasis based on numerous studies in solid tumors. Recently, evidence of their possible role in hematologic malignancies, specifically in the regulation of cellular adhesion, bone marrow homing, intracellular signaling, and stem cell dynamics in malignant hematologic cells has come to light. Many of these effects are facilitated by associations with CAMs and other receptors on the cell surface in TSPAN-enriched microdomains. This could suggest that TSPANs play an important role in mediating BMC in hematologic malignancies and could be used as therapeutic targets. In this review, we discuss TSPAN structure and function in hematologic cells, their interactions with different cell surface and signaling proteins, and possible ways to target/inhibit their effects.
© 2023 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current affiliation for A.Q. is Dr. Kiran C. Patel College of Allopathic Medicine, Nova Southeastern University, Fort Lauderdale, FL.
Figures




References
-
- Sidhu I, Barwe SP, Gopalakrishnapillai A. The extracellular matrix: a key player in the pathogenesis of hematologic malignancies. Blood Rev. 2021;48:100787. - PubMed
-
- Meads MB, Hazlehurst LA, Dalton WS. The bone marrow microenvironment as a tumor sanctuary and contributor to drug resistance. Clin Cancer Res. 2008;14(9):2519–2526. - PubMed
-
- Horejsi V, Vlcek C. Novel structurally distinct family of leucocyte surface glycoproteins including CD9, CD37, CD53 and CD63. FEBS Lett. 1991;288(1-2):1–4. - PubMed

