Exploring the landscape of Babesia bovis vaccines: progress, challenges, and opportunities
- PMID: 37563668
- PMCID: PMC10413621
- DOI: 10.1186/s13071-023-05885-z
Exploring the landscape of Babesia bovis vaccines: progress, challenges, and opportunities
Abstract
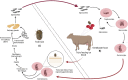
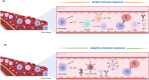
Bovine babesiosis, caused by different Babesia spp. such as B. bovis, B. bigemina, B. divergens, and B. major, is a global disease that poses a serious threat to livestock production. Babesia bovis infections are associated with severe disease and increased mortality in adult cattle, making it the most virulent agent of bovine babesiosis. Babesia bovis parasites undergo asexual reproduction within bovine red blood cells, followed by sexual reproduction within their tick vectors, which transmit the parasite transovarially. Current control methods, including therapeutic drugs (i.e., imidocarb) have been found to lead to drug resistance. Moreover, changing environmental factors add complexity to efficient parasite control. Understanding the fundamental biology, host immune responses, and host-parasite interactions of Babesia parasites is critical for developing next-generation vaccines to control acute disease and parasite transmission. This systematic review analyzed available research papers on vaccine development and the associated immune responses to B. bovis. We compiled and consolidated the reported vaccine strategies, considering the study design and rationale of each study, to provide a systematic review of knowledge and insights for further research. Thirteen studies published since 2014 (inclusive) represented various vaccine strategies developed against B. bovis such as subunit, live attenuated, and viral vector vaccines. Such strategies incorporated B. bovis proteins or whole live parasites with the latter providing the most effective prophylaxis against bovine babesiosis. Incorporating novel research approaches, such as "omics" will enhance our understanding of parasite vulnerabilities.
Keywords: Babesia bovis; Bovine babesiosis; Immune response; Vaccine development.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Unravelling the cellular and molecular pathogenesis of bovine babesiosis: is the sky the limit?Int J Parasitol. 2019 Feb;49(2):183-197. doi: 10.1016/j.ijpara.2018.11.002. Epub 2019 Jan 26. Int J Parasitol. 2019. PMID: 30690089 Free PMC article. Review.
-
Babesia bovis Enolase Is Expressed in Intracellular Merozoites and Contains B-Cell Epitopes That Induce Neutralizing Antibodies In Vitro.Vaccines (Basel). 2025 Jul 31;13(8):818. doi: 10.3390/vaccines13080818. Vaccines (Basel). 2025. PMID: 40872905
-
Immunogenicity and seroefficacy of pneumococcal conjugate vaccines: a systematic review and network meta-analysis.Health Technol Assess. 2024 Jul;28(34):1-109. doi: 10.3310/YWHA3079. Health Technol Assess. 2024. PMID: 39046101 Free PMC article.
-
Transgenic Babesia bovis lacking 6-Cys sexual-stage genes as the foundation for non-transmissible live vaccines against bovine babesiosis.Ticks Tick Borne Dis. 2019 Apr;10(3):722-728. doi: 10.1016/j.ttbdis.2019.01.006. Epub 2019 Jan 25. Ticks Tick Borne Dis. 2019. PMID: 30711475
-
Elucidating the functional role of the novel BdP50 protein and extracellular vesicles in the human erythrocyte infection by Babesia divergens.PLoS Negl Trop Dis. 2025 Aug 13;19(8):e0013401. doi: 10.1371/journal.pntd.0013401. eCollection 2025 Aug. PLoS Negl Trop Dis. 2025. PMID: 40802835 Free PMC article.
Cited by
-
Structural and antigenic characterization of Babesia Bovis HAP2 domains.Sci Rep. 2025 Mar 5;15(1):7781. doi: 10.1038/s41598-025-91359-4. Sci Rep. 2025. PMID: 40044720 Free PMC article.
-
Alternative and Complementary Approaches to Consider for Effective Babesia Vaccine Development.Pathogens. 2023 Sep 16;12(9):1166. doi: 10.3390/pathogens12091166. Pathogens. 2023. PMID: 37764974 Free PMC article.
-
A conserved motif in the immune-subdominant RAP-1 related antigen of Babesia bovis contains a B-cell epitope recognized by antibodies from protected cattle.Front Immunol. 2024 Apr 24;15:1380660. doi: 10.3389/fimmu.2024.1380660. eCollection 2024. Front Immunol. 2024. PMID: 38720894 Free PMC article.
References
-
- Kirupananthan R, Kamaral LCJ, Galhena GH, Perera KLN, Magamage MPS. Address the public health and food security concerns of babesiosis through molecular detection of Babesia bovis in suspected carrier cattle of selected localities in Sri Lanka. Procedia Food Sci. 2016;6:213–219. doi: 10.1016/j.profoo.2016.02.053. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

