Assessing Patient Adherence to and Engagement With Digital Interventions for Depression in Clinical Trials: Systematic Literature Review
- PMID: 37566447
- PMCID: PMC10457707
- DOI: 10.2196/43727
Assessing Patient Adherence to and Engagement With Digital Interventions for Depression in Clinical Trials: Systematic Literature Review
Abstract
Background: New approaches to the treatment of depression are necessary for patients who do not respond to current treatments or lack access to them because of barriers such as cost, stigma, and provider shortage. Digital interventions for depression are promising; however, low patient engagement could limit their effectiveness.
Objective: This systematic literature review (SLR) assessed how participant adherence to and engagement with digital interventions for depression have been measured in the published literature, what levels of adherence and engagement have been reported, and whether higher adherence and increased engagement are linked to increased efficacy.
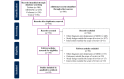
Methods: We focused on a participant population of adults (aged ≥18 years) with depression or major depressive disorder as the primary diagnosis and included clinical trials, feasibility studies, and pilot studies of digital interventions for treating depression, such as digital therapeutics. We screened 756 unique records from Ovid MEDLINE, Embase, and Cochrane published between January 1, 2000, and April 15, 2022; extracted data from and appraised the 94 studies meeting the inclusion criteria; and performed a primarily descriptive analysis. Otsuka Pharmaceutical Development & Commercialization, Inc (Princeton, New Jersey, United States) funded this study.
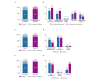
Results: This SLR encompassed results from 20,111 participants in studies using 47 unique web-based interventions (an additional 10 web-based interventions were not described by name), 15 mobile app interventions, 5 app-based interventions that are also accessible via the web, and 1 CD-ROM. Adherence was most often measured as the percentage of participants who completed all available modules. Less than half (44.2%) of the participants completed all the modules; however, the average dose received was 60.7% of the available modules. Although engagement with digital interventions was measured differently in different studies, it was most commonly measured as the number of modules completed, the mean of which was 6.4 (means ranged from 1.0 to 19.7) modules. The mean amount of time participants engaged with the interventions was 3.9 (means ranged from 0.7 to 8.4) hours. Most studies of web-based (34/45, 76%) and app-based (8/9, 89%) interventions found that the intervention group had substantially greater improvement for at least 1 outcome than the control group (eg, care as usual, waitlist, or active control). Of the 14 studies that investigated the relationship between engagement and efficacy, 9 (64%) found that increased engagement with digital interventions was significantly associated with improved participant outcomes. The limitations of this SLR include publication bias, which may overstate engagement and efficacy, and low participant diversity, which reduces the generalizability.
Conclusions: Patient adherence to and engagement with digital interventions for depression have been reported in the literature using various metrics. Arriving at more standardized ways of reporting adherence and engagement would enable more effective comparisons across different digital interventions, studies, and populations.
Keywords: adherence; depression; digital health; digital interventions; digital therapeutics; engagement; major depressive disorder; mobile health; mobile phone; systematic literature review.
©Ainslie Forbes, Madeline Rose Keleher, Michael Venditto, Faith DiBiasi. Originally published in the Journal of Medical Internet Research (https://www.jmir.org), 11.08.2023.
Conflict of interest statement
Conflicts of Interest: AF and FD are employees of Otsuka Pharmaceutical Development & Commercialization, Inc. MRK and MV are consultants for Otsuka Pharmaceutical Development & Commercialization, Inc.
Figures

References
-
- Awadalla S, Davies EB, Glazebrook C. A longitudinal cohort study to explore the relationship between depression, anxiety and academic performance among Emirati university students. BMC Psychiatry. 2020 Sep 11;20(1):448. doi: 10.1186/s12888-020-02854-z. https://bmcpsychiatry.biomedcentral.com/articles/10.1186/s12888-020-02854-z 10.1186/s12888-020-02854-z - DOI - PMC - PubMed
-
- Wallin E, Norlund F, Olsson EM, Burell G, Held C, Carlsson T. Treatment activity, user satisfaction, and experienced usability of internet-based cognitive behavioral therapy for adults with depression and anxiety after a myocardial infarction: mixed-methods study. J Med Internet Res. 2018 Mar 16;20(3):e87. doi: 10.2196/jmir.9690. https://www.jmir.org/2018/3/e87/ v20i3e87 - DOI - PMC - PubMed
-
- Saragoussi D, Christensen MC, Hammer-Helmich L, Rive B, Touya M, Haro JM. Long-term follow-up on health-related quality of life in major depressive disorder: a 2-year European cohort study. Neuropsychiatr Dis Treat. 2018 May 22;14:1339–50. doi: 10.2147/NDT.S159276. https://europepmc.org/abstract/MED/29872301 ndt-14-1339 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

