Chimeric antibody targeting unique epitope on onco-mucin16 reduces tumor burden in pancreatic and lung malignancies
- PMID: 37567918
- PMCID: PMC10421872
- DOI: 10.1038/s41698-023-00423-7
Chimeric antibody targeting unique epitope on onco-mucin16 reduces tumor burden in pancreatic and lung malignancies
Abstract
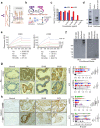
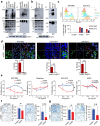
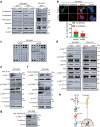
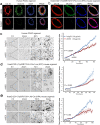
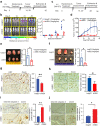
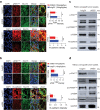
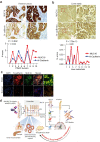
Aberrantly expressed onco-mucin 16 (MUC16) and its post-cleavage generated surface tethered carboxy-terminal (MUC16-Cter) domain are strongly associated with poor prognosis and lethality of pancreatic (PC) and non-small cell lung cancer (NSCLC). To date, most anti-MUC16 antibodies are directed towards the extracellular domain of MUC16 (CA125), which is usually cleaved and shed in the circulation hence obscuring antibody accessibility to the cancer cells. Herein, we establish the utility of targeting a post-cleavage generated, surface-tethered oncogenic MUC16 carboxy-terminal (MUC16-Cter) domain by using a novel chimeric antibody in human IgG1 format, ch5E6, whose epitope expression directly correlates with disease severity in both cancers. ch5E6 binds and interferes with MUC16-associated oncogenesis, suppresses the downstream signaling pFAK(Y397)/p-p70S6K(T389)/N-cadherin axis and exert antiproliferative effects in cancer cells, 3D organoids, and tumor xenografts of both PC and NSCLC. The robust clinical correlations observed between MUC16 and N-cadherin in patient tumors and metastatic samples imply ch5E6 potential in targeting a complex and significantly occurring phenomenon of epithelial to mesenchymal transition (EMT) associated with disease aggressiveness. Our study supports evaluating ch5E6 with standard-of-care drugs, to potentially augment treatment outcomes in malignancies inflicted with MUC16-associated poor prognosis.
© 2023. Nature Publishing Group UK.
Conflict of interest statement
S.K.B. is one of the co-founders of Sanguine Diagnostics and Therapeutics, Inc. A.K.G. has served as a consultant for AstraZeneca, Jazz Pharmaceuticals, Flagship Biosciences, G1 Therapeutics, Cardinal Health, Mirati Therapeutics, Beigene Ltd, Sanofi Genzyme and Regeneron Pharmaceuticals DSMB Chair for Y-mAbs Therapeutics outside the submitted work. The other authors declare no potential conflicts of interest.
Figures
Similar articles
-
Carboxyl-terminal domain of MUC16 imparts tumorigenic and metastatic functions through nuclear translocation of JAK2 to pancreatic cancer cells.Oncotarget. 2015 Mar 20;6(8):5772-87. doi: 10.18632/oncotarget.3308. Oncotarget. 2015. PMID: 25691062 Free PMC article.
-
Downregulation of cell surface CA125/MUC16 induces epithelial-to-mesenchymal transition and restores EGFR signalling in NIH:OVCAR3 ovarian carcinoma cells.Br J Cancer. 2011 Mar 15;104(6):989-99. doi: 10.1038/bjc.2011.34. Epub 2011 Feb 15. Br J Cancer. 2011. PMID: 21326240 Free PMC article.
-
Membrane proximal ectodomain cleavage of MUC16 occurs in the acidifying Golgi/post-Golgi compartments.Sci Rep. 2015 Jun 5;5:9759. doi: 10.1038/srep09759. Sci Rep. 2015. PMID: 26044153 Free PMC article.
-
[MUC16: The Novel Target for Tumor Therapy].Zhongguo Fei Ai Za Zhi. 2022 Jul 20;25(7):452-459. doi: 10.3779/j.issn.1009-3419.2022.101.31. Zhongguo Fei Ai Za Zhi. 2022. PMID: 35899441 Free PMC article. Review. Chinese.
-
Understanding the Unique Attributes of MUC16 (CA125): Potential Implications in Targeted Therapy.Cancer Res. 2015 Nov 15;75(22):4669-74. doi: 10.1158/0008-5472.CAN-15-1050. Epub 2015 Nov 2. Cancer Res. 2015. PMID: 26527287 Free PMC article. Review.
Cited by
-
MUC16: clinical targets with great potential.Clin Exp Med. 2024 May 17;24(1):101. doi: 10.1007/s10238-024-01365-5. Clin Exp Med. 2024. PMID: 38758220 Free PMC article. Review.
-
Mucins 3A and 3B Are Expressed in the Epithelium of Human Large Airway.Int J Mol Sci. 2023 Aug 31;24(17):13546. doi: 10.3390/ijms241713546. Int J Mol Sci. 2023. PMID: 37686350 Free PMC article.
-
Secretory Trefoil Factor 1 (TFF1) promotes gemcitabine resistance through chemokine receptor CXCR4 in Pancreatic Ductal Adenocarcinoma.Cancer Lett. 2024 Aug 28;598:217097. doi: 10.1016/j.canlet.2024.217097. Epub 2024 Jul 2. Cancer Lett. 2024. PMID: 38964729 Free PMC article.
-
MUC1 and MUC16: critical for immune modulation in cancer therapeutics.Front Immunol. 2024 Feb 1;15:1356913. doi: 10.3389/fimmu.2024.1356913. eCollection 2024. Front Immunol. 2024. PMID: 38361923 Free PMC article. Review.
-
Mesothelin-based CAR-T cells exhibit potent antitumor activity against ovarian cancer.J Transl Med. 2024 Apr 18;22(1):367. doi: 10.1186/s12967-024-05174-y. J Transl Med. 2024. PMID: 38637885 Free PMC article.
References
-
- Mullard A. FDA approves 100th monoclonal antibody product. Nat. Rev. Drug Discov. 2021;20:491–495. - PubMed
Grants and funding
- U01 CA200466/CA/NCI NIH HHS/United States
- R01 CA247471/CA/NCI NIH HHS/United States
- R01 CA228524/CA/NCI NIH HHS/United States
- R01 CA263575/CA/NCI NIH HHS/United States
- U01 CA213862/CA/NCI NIH HHS/United States
- R01 CA256973/CA/NCI NIH HHS/United States
- R01 CA273349/CA/NCI NIH HHS/United States
- U01 CA210240/CA/NCI NIH HHS/United States
- R01 CA210637/CA/NCI NIH HHS/United States
- P01 CA217798/CA/NCI NIH HHS/United States
- R01 CA206444/CA/NCI NIH HHS/United States
- R21 CA267582/CA/NCI NIH HHS/United States
- R01 CA273319/CA/NCI NIH HHS/United States
- R44 DK117472/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

