Establishment and Characterization of Mild Atopic Dermatitis in the DNCB-Induced Mouse Model
- PMID: 37569701
- PMCID: PMC10418750
- DOI: 10.3390/ijms241512325
Establishment and Characterization of Mild Atopic Dermatitis in the DNCB-Induced Mouse Model
Abstract
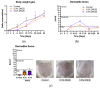
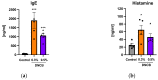
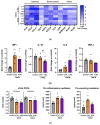
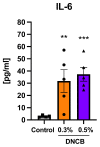
In dermatological research, 2,4-dinitrochlorbenzene (DNCB)-induced atopic dermatitis (AD) is a standard model as it displays many disease-associated characteristics of human AD. However, the reproducibility of the model is challenging due to the lack of information regarding the methodology and the description of the phenotype and endotype of the mimicked disease. In this study, a DNCB-induced mouse model was established with a detailed procedure description and classification of the AD human-like skin type. The disease was induced with 1% DNCB in the sensitization phase and repeated applications of 0.3% and 0.5% DNCB in the challenging phase which led to a mild phenotype of AD eczema. Pathophysiological changes of the dorsal skin were measured: thickening of the epidermis and dermis, altered skin barrier proteins, increased TH1 and TH2 cytokine expression, a shift in polyunsaturated fatty acids, increased pro-resolving and inflammatory mediator formation, and dysregulated inflammation-associated gene expression. A link to type I allergy reactions was evaluated by increased mast cell infiltration into the skin accompanied by elevated IgE and histamine levels in plasma. As expected for mild AD, no systemic inflammation was observed. In conclusion, this experimental setup demonstrates many features of a mild human-like extrinsic AD in murine skin.
Keywords: DNCB; atopic dermatitis mouse model; extrinsic; mild atopic dermatitis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Asher M.I., Montefort S., Björkstén B., Lai C.K., Strachan D.P., Weiland S.K., Williams H. Worldwide Time Trends in the Prevalence of Symptoms of Asthma, Allergic Rhinoconjunctivitis, and Eczema in Childhood: ISAAC Phases One and Three Repeat Multicountry Cross-Sectional Surveys. Lancet. 2006;368:733–743. doi: 10.1016/S0140-6736(06)69283-0. - DOI - PubMed
-
- Gittler J.K., Shemer A., Suárez-Fariñas M., Fuentes-Duculan J., Gulewicz K.J., Wang C.Q.F., Mitsui H., Cardinale I., de Guzman Strong C., Krueger J.G., et al. Progressive Activation of TH2/TH22 Cytokines and Selective Epidermal Proteins Characterizes Acute and Chronic Atopic Dermatitis. J. Allergy Clin. Immunol. 2012;130:1344–1354. doi: 10.1016/j.jaci.2012.07.012. - DOI - PMC - PubMed
-
- Burgess J.A., Dharmage S.C., Byrnes G.B., Matheson M.C., Gurrin L.C., Wharton C.L., Johns D.P., Abramson M.J., Hopper J.L., Walters E.H. Childhood Eczema and Asthma Incidence and Persistence: A Cohort Study from Childhood to Middle Age. J. Allergy Clin. Immunol. 2008;122:280–285. doi: 10.1016/j.jaci.2008.05.018. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

