Changes in the Mitochondria in the Aging Process-Can α-Tocopherol Affect Them?
- PMID: 37569829
- PMCID: PMC10419829
- DOI: 10.3390/ijms241512453
Changes in the Mitochondria in the Aging Process-Can α-Tocopherol Affect Them?
Abstract
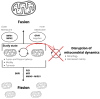
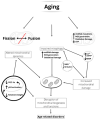
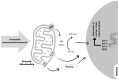
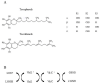
Aerobic organisms use molecular oxygen in several reactions, including those in which the oxidation of substrate molecules is coupled to oxygen reduction to produce large amounts of metabolic energy. The utilization of oxygen is associated with the production of ROS, which can damage biological macromolecules but also act as signaling molecules, regulating numerous cellular processes. Mitochondria are the cellular sites where most of the metabolic energy is produced and perform numerous physiological functions by acting as regulatory hubs of cellular metabolism. They retain the remnants of their bacterial ancestors, including an independent genome that encodes part of their protein equipment; they have an accurate quality control system; and control of cellular functions also depends on communication with the nucleus. During aging, mitochondria can undergo dysfunctions, some of which are mediated by ROS. In this review, after a description of how aging affects the mitochondrial quality and quality control system and the involvement of mitochondria in inflammation, we report information on how vitamin E, the main fat-soluble antioxidant, can protect mitochondria from age-related changes. The information in this regard is scarce and limited to some tissues and some aspects of mitochondrial alterations in aging. Improving knowledge of the effects of vitamin E on aging is essential to defining an optimal strategy for healthy aging.
Keywords: ROS production; inflamm-aging; mitochondrial dynamics; mtDNA; oxidative damage; vitamin E; vitamin E metabolites.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Vitamin E Supplementation and Mitochondria in Experimental and Functional Hyperthyroidism: A Mini-Review.Nutrients. 2019 Dec 1;11(12):2900. doi: 10.3390/nu11122900. Nutrients. 2019. PMID: 31805673 Free PMC article. Review.
-
Transcription Factor Nrf2 and Mitochondria - Friends or Foes in the Regulation of Aging Rate.Biochemistry (Mosc). 2022 Dec;87(12):1477-1486. doi: 10.1134/S0006297922120057. Biochemistry (Mosc). 2022. PMID: 36717441 Review.
-
Oxidative stress and protein aggregation during biological aging.Exp Gerontol. 2001 Sep;36(9):1539-50. doi: 10.1016/s0531-5565(01)00139-5. Exp Gerontol. 2001. PMID: 11525876 Review.
-
Influence of aging and long-term caloric restriction on oxygen radical generation and oxidative DNA damage in rat liver mitochondria.Free Radic Biol Med. 2002 May 1;32(9):882-9. doi: 10.1016/s0891-5849(02)00773-6. Free Radic Biol Med. 2002. PMID: 11978489
-
Melatonin-mitochondria interplay in health and disease.Curr Top Med Chem. 2011;11(2):221-40. doi: 10.2174/156802611794863517. Curr Top Med Chem. 2011. PMID: 21244359 Review.
Cited by
-
Photobiomodulation in the aging brain: a systematic review from animal models to humans.Geroscience. 2024 Dec;46(6):6583-6623. doi: 10.1007/s11357-024-01231-y. Epub 2024 Jun 11. Geroscience. 2024. PMID: 38861125 Free PMC article.
-
Effects of hypervitaminosis C and D on spermatogenesis through CDKN1B modulation in adult male mice: biochemical and immunohistochemical studies.J Mol Histol. 2025 Jun 21;56(4):204. doi: 10.1007/s10735-025-10483-7. J Mol Histol. 2025. PMID: 40542892 No abstract available.
-
The cardiac response of the goldfish Carassius auratus to environmental hypoxia: from hemodynamics to mitochondria.Fish Physiol Biochem. 2025 Jan 24;51(1):36. doi: 10.1007/s10695-025-01452-8. Fish Physiol Biochem. 2025. PMID: 39849270 Free PMC article.
References
-
- Williams G.C. Pleiotropy, natural selection and the evolution of senescence. Evolution. 1957;11:398–411. doi: 10.2307/2406060. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

