Global commercialization and research of veterinary vaccines against Pasteurella multocida: 2015-2022 technological surveillance
- PMID: 37576757
- PMCID: PMC10420726
- DOI: 10.14202/vetworld.2023.946-956
Global commercialization and research of veterinary vaccines against Pasteurella multocida: 2015-2022 technological surveillance
Abstract
Background and aim: Pasteurella multocida can infect a multitude of wild and domesticated animals, bacterial vaccines have become a crucial tool in combating antimicrobial resistance (AMR) in animal production. The study aimed to evaluate the current status and scientific trends related to veterinary vaccines against Pasteurella multocida during the 2015-2022 period.
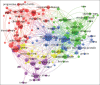
Material and methods: The characteristics of globally marketed vaccines were investigated based on the official websites of 22 pharmaceutical companies. VOSviewer® 1.6.18 was used to visualize networks of coauthorship and cooccurrence of keywords from papers published in English and available in Scopus.
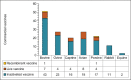
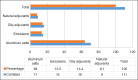
Results: Current commercial vaccines are mostly inactivated (81.7%), adjuvanted in aluminum hydroxide (57.8%), and designed to immunize cattle (33.0%). Investigational vaccines prioritize the inclusion of attenuated strains, peptide fragments, recombinant proteins, DNA as antigens, aluminum compounds as adjuvants and poultry as the target species.
Conclusion: Despite advances in genetic engineering and biotechnology, there will be no changes in the commercial dominance of inactivated and aluminum hydroxide-adjuvanted vaccines in the short term (3-5 years). The future prospects for bacterial vaccines in animal production are promising, with advancements in vaccine formulation and genetic engineering, they have the potential to improve the sustainability of the industry. It is necessary to continue with the studies to improve the efficacy of the vaccines and their availability.
Keywords: Pasteurellamultocida; adjuvant; strain; technological surveillance; vaccine.
Copyright: © Domínguez-Odio and Delgado.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Harper M, Boyce J.D, Adler B. Pasteurella multocida pathogenesis:125 years after Pasteur. FEMS Microbiol. Lett. 2006;265(1):1–10. - PubMed
-
- Shivachandra S.B, Viswas K.N, Kumar A.A. A review of haemorrhagic septicaemia in catte and buffalo. Anim. Health Res. 2011;12(1):67–82. - PubMed
-
- Pors S.E, Hansen M.S, Christensen H, Jensen H.E, Petersen A, Bisgaard M. Genetic diversity and associated pathology of Pasteurella multocida isolated from porcine pneumonia. Vet. Microbiol. 2011;150(3–4):354–361. - PubMed
LinkOut - more resources
Full Text Sources
