Preclinical in vitro evaluation of implantable materials: conventional approaches, new models and future directions
- PMID: 37576997
- PMCID: PMC10416115
- DOI: 10.3389/fbioe.2023.1193204
Preclinical in vitro evaluation of implantable materials: conventional approaches, new models and future directions
Abstract
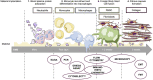
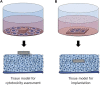
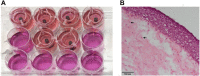
Nowadays, implants and prostheses are widely used to repair damaged tissues or to treat different diseases, but their use is associated with the risk of infection, inflammation and finally rejection. To address these issues, new antimicrobial and anti-inflammatory materials are being developed. Aforementioned materials require their thorough preclinical testing before clinical applications can be envisaged. Although many researchers are currently working on new in vitro tissues for drug screening and tissue replacement, in vitro models for evaluation of new biomaterials are just emerging and are extremely rare. In this context, there is an increased need for advanced in vitro models, which would best recapitulate the in vivo environment, limiting animal experimentation and adapted to the multitude of these materials. Here, we overview currently available preclinical methods and models for biological in vitro evaluation of new biomaterials. We describe several biological tests used in biocompatibility assessment, which is a primordial step in new material's development, and discuss existing challenges in this field. In the second part, the emphasis is made on the development of new 3D models and approaches for preclinical evaluation of biomaterials. The third part focuses on the main parameters to consider to achieve the optimal conditions for evaluating biocompatibility; we also overview differences in regulations across different geographical regions and regulatory systems. Finally, we discuss future directions for the development of innovative biomaterial-related assays: in silico models, dynamic testing models, complex multicellular and multiple organ systems, as well as patient-specific personalized testing approaches.
Keywords: 3D models; biocompatibility; biomaterials; implants; organoids.
Copyright © 2023 Frisch, Clavier, Belhamdi, Vrana, Lavalle, Frisch, Heurtault and Gribova.
Conflict of interest statement
Authors AB, NV, and PL are employed by the company SPARTHA Medical. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Akiva A., Melke J., Ansari S., Liv N., van der Meijden R., van Erp M., et al. (2021). An organoid for woven bone. Adv. Funct. Mater. 31 (17), 2010524. 10.1002/adfm.202010524 - DOI
Publication types
LinkOut - more resources
Full Text Sources

