This is a preprint.
Cell autonomous role of border associated macrophages in ApoE4 neurovascular dysfunction and susceptibility to white matter injury
- PMID: 37577565
- PMCID: PMC10418550
- DOI: 10.21203/rs.3.rs-3222611/v1
Cell autonomous role of border associated macrophages in ApoE4 neurovascular dysfunction and susceptibility to white matter injury
Update in
-
A cell-autonomous role for border-associated macrophages in ApoE4 neurovascular dysfunction and susceptibility to white matter injury.Nat Neurosci. 2024 Nov;27(11):2138-2151. doi: 10.1038/s41593-024-01757-6. Epub 2024 Sep 18. Nat Neurosci. 2024. PMID: 39294490 Free PMC article.
Abstract
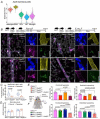
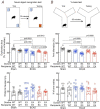
Apolipoprotein-E4 (ApoE4), the strongest genetic risk factor for sporadic Alzheimer's disease, is also a risk factor for microvascular pathologies leading to cognitive impairment, particularly subcortical white matter injury. These effects have been attributed to alterations in the regulation of the brain blood supply, but the cellular source of ApoE4 and the underlying mechanisms remain unclear. In mice expressing human ApoE3 or ApoE4 we report that border associated macrophages (BAM), myeloid cells closely apposed to neocortical microvessels, are both the source and the target of the ApoE4 mediating the neurovascular dysfunction through reactive oxygen species. ApoE4 in BAM is solely responsible for the increased susceptibility to oligemic white matter damage in ApoE4 mice and is sufficient to enhance damage in ApoE3 mice. The data unveil a new aspect of BAM pathobiology and highlight a previously unrecognized cell autonomous role of BAM in the neurovascular dysfunction of ApoE4 with potential therapeutic implications.
Figures





References
-
- Zlokovic B.V., et al. Vascular contributions to cognitive impairment and dementia (VCID): A report from the 2018 National Heart, Lung, and Blood Institute and National Institute of Neurological Disorders and Stroke Workshop. Alzheimer’s & dementia : the journal of the Alzheimer’s Association 16, 1714–1733 (2020). - PubMed
-
- Gouw A.A., et al. Heterogeneity of white matter hyperintensities in Alzheimer’s disease: post-mortem quantitative MRI and neuropathology. Brain 131, 3286–3298 (2008). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

