Diagnostic accuracy of risk assessment and fecal immunochemical test in colorectal cancer screening: Results from a population-based program and meta-analysis
- PMID: 37578430
- PMCID: PMC10524075
- DOI: 10.1002/cam4.6399
Diagnostic accuracy of risk assessment and fecal immunochemical test in colorectal cancer screening: Results from a population-based program and meta-analysis
Abstract
Background: Fecal immunochemical test (FIT) is a commonly used initial test for colorectal cancer (CRC) screening. Parallel use of FIT with risk assessment (RA) could improve the detection of non-bleeding lesions, but at the expense of compromising sensitivity. In this study, we evaluated the accuracy of FIT and/or RA in the Shanghai CRC screening program, and systematically reviewed the relevant evaluations worldwide.
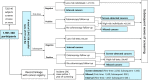
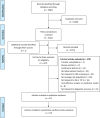
Methods: RA and 2-specimen FIT were used in parallel in the Shanghai screening program, followed by a colonoscopy among those with positive results. Sensitivity, specificity, detection rate of CRC, positive predictive value (PPV), and other measures with their 95% confident intervals were calculated for each type of tests and several assumed combined tests. We further searched PubMed, Embase, Web of Science, and Cochrane Library for relevant studies published in English up to January 5, 2022.
Results: By the end of 2019, a total of 1,901,360 participants of the screening program completed 3,045,108 tests, with 1,901,360 first-time tests and 1,143,748 subsequent tests. Parallel use of RA and 2-specimen FIT achieved a sensitivity of 0.78 (0.77-0.80), a specificity of 0.78 (0.78-0.78), PPV of 0.89% (0.86-0.92), and a detection rate of 1.99 (1.93-2.05) for CRC per 1000 among participants enrolled in the first screening round, and performed similarly among those who participated for several times. A meta-analysis of 103 published observational studies demonstrated a higher sensitivity [0.76 (0.36, 0.94)] but a much lower specificity [0.59 (0.28, 0.85)] of parallel use of RA and FIT for detecting CRC in average-risk populations than in our subjects. One-specimen FIT, the most commonly used initial test, had a pooled specificity comparable to the Shanghai screening program (0.92 vs. 0.91), but a much higher pooled sensitivity (0.76 vs. 0.57).
Conclusion: Our results indicate the limitation of FIT only as an initial screening test for CRC in Chinese populations, and highlight the higher sensitivity of parallel use of RA and FIT. Attempts should be made to optimize RA to improve effectiveness of screening in the populations.
Keywords: colorectal cancer; diagnostic accuracy; fecal immunochemical test; risk assessment; screening.
© 2023 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
All authors declare no competing interests.
Figures

Similar articles
-
Participant-Related Risk Factors for False-Positive and False-Negative Fecal Immunochemical Tests in Colorectal Cancer Screening: Systematic Review and Meta-Analysis.Am J Gastroenterol. 2018 Dec;113(12):1778-1787. doi: 10.1038/s41395-018-0212-7. Epub 2018 Aug 29. Am J Gastroenterol. 2018. PMID: 30158713 Free PMC article.
-
Fecal occult blood test for colorectal cancer screening: an evidence-based analysis.Ont Health Technol Assess Ser. 2009;9(10):1-40. Epub 2009 Sep 1. Ont Health Technol Assess Ser. 2009. PMID: 23074514 Free PMC article.
-
The second round of the Dutch colorectal cancer screening program: Impact of an increased fecal immunochemical test cut-off level on yield of screening.Int J Cancer. 2020 Aug 15;147(4):1098-1106. doi: 10.1002/ijc.32839. Epub 2020 Jan 9. Int J Cancer. 2020. PMID: 31853977 Free PMC article.
-
Yield and Effectiveness of 2-Sample Fecal Immunochemical Test-Based Screening Program for Colorectal Cancer.Clin Gastroenterol Hepatol. 2025 Jun;23(7):1238-1246.e6. doi: 10.1016/j.cgh.2024.10.010. Epub 2024 Nov 6. Clin Gastroenterol Hepatol. 2025. PMID: 39510222
-
Screening for Colorectal Cancer: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force.JAMA. 2021 May 18;325(19):1978-1998. doi: 10.1001/jama.2021.4417. JAMA. 2021. PMID: 34003220
References
-
- International Agency for Research on Cancer . Colorectal Cancer: Estimated Incidence, Mortality and Prevalence Worldwide in 2020. World Health Organization; 2021.
-
- Shaukat A, Kahi CJ, Burke CA, Rabeneck L, Sauer BG, Rex DK. ACG clinical guidelines: colorectal cancer screening 2021. Am J Gastroenterol. 2021;116(3):458‐479. - PubMed
-
- Lertkhachonsuk AA, Yip CH, Khuhaprema T, et al. Cancer prevention in Asia: resource‐stratified guidelines from the Asian Oncology Summit 2013. Lancet Oncol. 2013;14(12):e497‐e507. - PubMed
-
- Jenkins MA, Ait Ouakrim D, Boussioutas A, et al. Revised Australian national guidelines for colorectal cancer screening: family history. Med J Aust. 2018;209(10):455‐460. - PubMed
-
- Sung JJ, Ng SC, Chan FK, et al. An updated Asia Pacific Consensus Recommendations on colorectal cancer screening. Gut. 2015;64(1):121‐132. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

