WASF3 disrupts mitochondrial respiration and may mediate exercise intolerance in myalgic encephalomyelitis/chronic fatigue syndrome
- PMID: 37579159
- PMCID: PMC10450651
- DOI: 10.1073/pnas.2302738120
WASF3 disrupts mitochondrial respiration and may mediate exercise intolerance in myalgic encephalomyelitis/chronic fatigue syndrome
Abstract
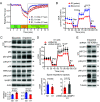
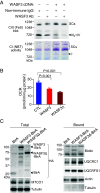
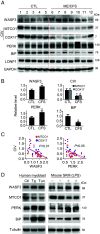
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is characterized by various disabling symptoms including exercise intolerance and is diagnosed in the absence of a specific cause, making its clinical management challenging. A better understanding of the molecular mechanism underlying this apparent bioenergetic deficiency state may reveal insights for developing targeted treatment strategies. We report that overexpression of Wiskott-Aldrich Syndrome Protein Family Member 3 (WASF3), here identified in a 38-y-old woman suffering from long-standing fatigue and exercise intolerance, can disrupt mitochondrial respiratory supercomplex formation and is associated with endoplasmic reticulum (ER) stress. Increased expression of WASF3 in transgenic mice markedly decreased their treadmill running capacity with concomitantly impaired respiratory supercomplex assembly and reduced complex IV levels in skeletal muscle mitochondria. WASF3 induction by ER stress using endotoxin, well known to be associated with fatigue in humans, also decreased skeletal muscle complex IV levels in mice, while decreasing WASF3 levels by pharmacologic inhibition of ER stress improved mitochondrial function in the cells of the patient with chronic fatigue. Expanding on our findings, skeletal muscle biopsy samples obtained from a cohort of patients with ME/CFS showed increased WASF3 protein levels and aberrant ER stress activation. In addition to revealing a potential mechanism for the bioenergetic deficiency in ME/CFS, our study may also provide insights into other disorders associated with fatigue such as rheumatic diseases and long COVID.
Keywords: ER stress; WASF3; fatigue; mitochondria; supercomplex.
Conflict of interest statement
The authors declare no competing interest.
Figures



References
-
- Bateman L., et al. , Myalgic encephalomyelitis/chronic fatigue syndrome: Essentials of diagnosis and management. Mayo. Clin. Proc. 96, 2861–2878 (2021). - PubMed
-
- Institute of Medicine (U.S.), “Committee on the diagnostic criteria for myalgic encephalomyelitis/chronic fatigue syndrome & Institute of Medicine (U.S.). Board on the Health of Select Populations” in Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness (The National Academies Press, Washington, D.C., 2015), p. XXI, 282 pp.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

