A Novel Near-Infrared Fluorescence Probe THK-565 Enables In Vivo Detection of Amyloid Deposits in Alzheimer's Disease Mouse Model
- PMID: 37580462
- PMCID: PMC10728248
- DOI: 10.1007/s11307-023-01843-4
A Novel Near-Infrared Fluorescence Probe THK-565 Enables In Vivo Detection of Amyloid Deposits in Alzheimer's Disease Mouse Model
Abstract
Purpose: Noninvasive imaging of protein aggregates in the brain is critical for the early diagnosis, disease monitoring, and evaluation of the effectiveness of novel therapies for Alzheimer's disease (AD). Near-infrared fluorescence (NIRF) imaging with specific probes is a promising technique for the in vivo detection of protein deposits without radiation exposure. Comprehensive screening of fluorescent compounds identified a novel compound, THK-565, for the in vivo imaging of amyloid-β (Aβ) deposits in the mouse brain. This study assessed whether THK-565 could detect amyloid-β deposits in vivo in the AD mouse model.
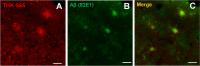
Procedures: The fluorescent properties of THK-565 were evaluated in the presence and absence of Aβ fibrils. APP knock-in (APP-KI) mice were used as an animal model of AD. In vivo NIRF images were acquired after the intravenous administration of THK-565 and THK-265 in mice. The binding selectivity of THK-565 to Aβ was evaluated using brain slices obtained from these mouse models.
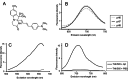
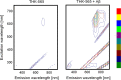
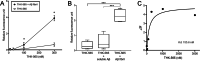
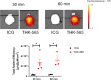
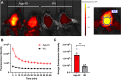
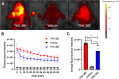
Results: The fluorescence intensity of the THK-565 solution substantially increased by mixing with Aβ fibrils. The maximum emission wavelength of the complex of THK-565 and Aβ fibrils was 704 nm, which was within the optical window range. THK-565 selectively bound to amyloid deposits in brain sections of APP-KI mice After the intravenous administration of THK-565, the fluorescence signal in the head of APP-KI mice was significantly higher than that of wild-type mice and higher than that after administration of THK-265. Ex vivo analysis confirmed that the THK-565 signal corresponded to Aβ immunostaining in the brain sections of these mice.
Conclusions: A novel NIRF probe, THK-565, enabled the in vivo detection of Aβ deposits in the brains of the AD mouse model, suggesting that NIRF imaging with THK-565 could non-invasively assess disease-specific pathology in AD.
Keywords: Alzheimer’s disease; Amyloid; Fluorescence; Imaging.
© 2023. The Author(s).
Conflict of interest statement
Drs Furumoto, Kudo, and Okamura have a patent pending for the technology described in this manuscript. No other potential conflicts of interest relevant to this article exist.
Figures

Similar articles
-
In vivo detection of amyloid plaques in the mouse brain using the near-infrared fluorescence probe THK-265.J Alzheimers Dis. 2011;23(1):37-48. doi: 10.3233/JAD-2010-100270. J Alzheimers Dis. 2011. PMID: 20930313
-
Efficient near-infrared in vivo imaging of amyoid-β deposits in Alzheimer's disease mouse models.J Alzheimers Dis. 2012;30(3):651-64. doi: 10.3233/JAD-2012-112168. J Alzheimers Dis. 2012. PMID: 22460331
-
Engineering of donor-acceptor-donor curcumin analogues as near-infrared fluorescent probes for in vivo imaging of amyloid-β species.Theranostics. 2022 Apr 4;12(7):3178-3195. doi: 10.7150/thno.68679. eCollection 2022. Theranostics. 2022. PMID: 35547754 Free PMC article.
-
[Development of SPECT Probes for In Vivo Imaging of β-Amyloid and Tau Aggregates in the Alzheimer's Disease Brain].Yakugaku Zasshi. 2017;137(11):1361-1365. doi: 10.1248/yakushi.17-00156. Yakugaku Zasshi. 2017. PMID: 29093372 Review. Japanese.
-
Versatile fluorescent probes for near-infrared imaging of amyloid-β species in Alzheimer's disease mouse model.J Mater Chem B. 2019 Mar 28;7(12):1986-1995. doi: 10.1039/c9tb00161a. Epub 2019 Mar 1. J Mater Chem B. 2019. PMID: 32254802 Review.
Cited by
-
Ligands for Protein Fibrils of Amyloid-β, α-Synuclein, and Tau.Chem Rev. 2025 Jun 11;125(11):5282-5348. doi: 10.1021/acs.chemrev.4c00838. Epub 2025 May 6. Chem Rev. 2025. PMID: 40327808 Free PMC article. Review.
-
Visualizing alpha-synuclein and iron deposition in M83 mouse model of Parkinson's disease in vivo.Brain Pathol. 2024 Nov;34(6):e13288. doi: 10.1111/bpa.13288. Epub 2024 Jul 9. Brain Pathol. 2024. PMID: 38982662 Free PMC article.
References
-
- Zwan MD, Okamura N, Fodero-Tavoletti MT et al (2014) Voyage au bout de la nuit: Aβ and tau imaging in dementias. Q J Nucl Med Mol Imaging 58:398–412 - PubMed
-
- Okamura N, Harada R (2022) PET imaging of amyloid and tau in Alzheimer’s disease. In Mori N (ed.) Aging Mechanisms II : Longevity, Metabolism, and Brain Aging. Singapore: Springer Nature Singapore, 307–323
-
- Hintersteiner M, Enz A, Frey P et al (2005) In vivo detection of amyloid-β deposits by near-infrared imaging using an oxazine-derivative probe. Nat Biotechnol 23:577–583 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

