Amelioration of non-alcoholic fatty liver disease by targeting adhesion G protein-coupled receptor F1 (Adgrf1)
- PMID: 37580962
- PMCID: PMC10427146
- DOI: 10.7554/eLife.85131
Amelioration of non-alcoholic fatty liver disease by targeting adhesion G protein-coupled receptor F1 (Adgrf1)
Erratum in
-
Correction: Amelioration of non-alcoholic fatty liver disease by targeting adhesion G protein-coupled receptor F1 (Adgrf1).Elife. 2024 Jan 22;13:e96183. doi: 10.7554/eLife.96183. Elife. 2024. PMID: 38252482 Free PMC article.
Abstract
Background: Recent research has shown that the adhesion G protein-coupled receptor F1 (Adgrf1; also known as GPR110; PGR19; KPG_012; hGPCR36) is an oncogene. The evidence is mainly based on high expression of Adgrf1 in numerous cancer types, and knockdown Adgrf1 can reduce the cell migration, invasion, and proliferation. Adgrf1 is, however, mostly expressed in the liver of healthy individuals. The function of Adgrf1 in liver has not been revealed. Interestingly, expression level of hepatic Adgrf1 is dramatically decreased in obese subjects. Here, the research examined whether Adgrf1 has a role in liver metabolism.
Methods: We used recombinant adeno-associated virus-mediated gene delivery system, and antisense oligonucleotide was used to manipulate the hepatic Adgrf1 expression level in diet-induced obese mice to investigate the role of Adgrf1 in hepatic steatosis. The clinical relevance was examined using transcriptome profiling and archived biopsy specimens of liver tissues from non-alcoholic fatty liver disease (NAFLD) patients with different degree of fatty liver.
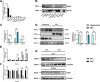
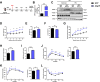
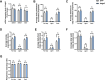
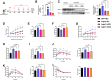
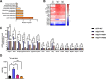
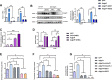
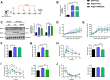
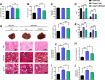
Results: The expression of Adgrf1 in the liver was directly correlated to fat content in the livers of both obese mice and NAFLD patients. Stearoyl-coA desaturase 1 (Scd1), a crucial enzyme in hepatic de novo lipogenesis, was identified as a downstream target of Adgrf1 by RNA-sequencing analysis. Treatment with the liver-specific Scd1 inhibitor MK8245 and specific shRNAs against Scd1 in primary hepatocytes improved the hepatic steatosis of Adgrf1-overexpressing mice and lipid profile of hepatocytes, respectively.
Conclusions: These results indicate Adgrf1 regulates hepatic lipid metabolism through controlling the expression of Scd1. Downregulation of Adgrf1 expression can potentially serve as a protective mechanism to stop the overaccumulation of fat in the liver in obese subjects. Overall, the above findings not only reveal a new mechanism regulating the progression of NAFLD, but also proposed a novel therapeutic approach to combat NAFLD by targeting Adgrf1.
Funding: This work was supported by the National Natural Science Foundation of China (81870586), Area of Excellence (AoE/M-707/18), and General Research Fund (15101520) to CMW, and the National Natural Science Foundation of China (82270941, 81974117) to SJ.
Keywords: SCD1; cell biology; fatty liver; gpr110; human; lipid metabolism; medicine; mouse; obesity.
Plain language summary
Being overweight or obese increases the risk of developing numerous medical conditions including non-alcoholic fatty liver disease (NAFLD), where excess fat accumulates in the liver. NAFLD is a major global health issue affecting about 25% of the world's population and, if left untreated, can lead to liver inflammation as well as serious complications such as type 2 diabetes, heart disease, and liver cancer. Currently, there are no medications which specifically treat NFALD. Instead, only medications which help to manage the associated health complications are available. Therefore, a better understanding of NFALD is required to help to develop new strategies for diagnosing and treating the progression of this disease. A family of proteins known as GPCRs have crucial roles in regulating various bodily processes and are therefore commonly targeted for the treatment of disease. By identifying the GPCRs specifically involved in liver fat accumulation, new treatments for NFALD could be identified. Previous studies identified a GPCR known as Adgrf1 that is mainly found in liver cells, but its role remained unclear. To investigate the function of Adgrf1 in the liver, Wu et al. studied obese mice and human patients with NAFLD. The experiments showed that elevated levels of Adgrf1 in human and mouse livers led to increased fat accumulation. On the other hand, livers with lower levels of Adgrf1 exhibited reduced fat levels. A technique called RNA sequencing revealed that Adgrf1 induces expression of enzymes involved in fat synthesis, including a key regulator called Scd1. Treating mice with high levels of liver fat with molecules that inhibit Scd1 decreased the symptoms of Adgrf1-mediated fatty liver disease. These findings suggest therapies that decrease the levels of Adgrf1 may help to stop too much fat accumulating in the liver of human patients who are at risk of developing NAFLD. Further research is needed to confirm the effectiveness and safety of targeting Adgrf1 in humans and to develop suitable candidate drugs for the task.
© 2023, Wu et al.
Conflict of interest statement
MW, CW has filed the following provisional patent: "Application of GPR110 as target for treating metabolic diseases"; US Provisional Filing Ref No. 63/370,948. The author has no other competing interests to declare, TL, LL, JS, CD, KC, XL, JL, PL, AX No competing interests declared, SY is affiliated with Ionis Pharmaceuticals, and holds stock at Ionis. The author has no other competing interests to declare
Figures



References
-
- Abdulkareem NM, Bhat R, Qin L, Vasaikar S, Gopinathan A, Mitchell T, Shea MJ, Nanda S, Thangavel H, Zhang B, De Angelis C, Schiff R, Trivedi MV. A novel role of ADGRF1 (GPR110) in promoting cellular quiescence and chemoresistance in human epidermal growth factor receptor 2-positive breast cancer. FASEB Journal. 2021;35:e21719. doi: 10.1096/fj.202100070R. - DOI - PMC - PubMed
-
- Ahrens M, Ammerpohl O, von Schönfels W, Kolarova J, Bens S, Itzel T, Teufel A, Herrmann A, Brosch M, Hinrichsen H, Erhart W, Egberts J, Sipos B, Schreiber S, Häsler R, Stickel F, Becker T, Krawczak M, Röcken C, Siebert R, Schafmayer C, Hampe J. DNA methylation analysis in nonalcoholic fatty liver disease suggests distinct disease-specific and remodeling signatures after bariatric surgery. Cell Metabolism. 2013;18:296–302. doi: 10.1016/j.cmet.2013.07.004. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

