Unleashing T cell anti-tumor immunity: new potential for 5-Nonloxytryptamine as an agent mediating MHC-I upregulation in tumors
- PMID: 37582744
- PMCID: PMC10426104
- DOI: 10.1186/s12943-023-01833-8
Unleashing T cell anti-tumor immunity: new potential for 5-Nonloxytryptamine as an agent mediating MHC-I upregulation in tumors
Abstract
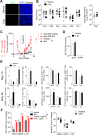
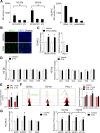
Background: New therapies are urgently needed in melanoma, particularly in late-stage patients not responsive to immunotherapies and kinase inhibitors. To uncover novel potentiators of T cell anti-tumor immunity, we carried out an ex vivo pharmacological screen and identified 5-Nonyloxytryptamine (5-NL), a serotonin agonist, as increasing the ability of T cells to target tumor cells.
Methods: The pharmacological screen utilized lymphocytic choriomeningitis virus (LCMV)-primed splenic T cells and melanoma B16.F10 cells expressing the LCMV gp33 CTL epitope. In vivo tumor growth in C57BL/6 J and NSG mice, in vivo antibody depletion, flow cytometry, immunoblot, CRISPR/Cas9 knockout, histological and RNA-Seq analyses were used to decipher 5-NL's immunomodulatory effects in vitro and in vivo.
Results: 5-NL delayed tumor growth in vivo and the phenotype was dependent on the hosts' immune system, specifically CD8+ T cells. 5-NL's pro-immune effects were not directly consequential to T cells. Rather, 5-NL upregulated antigen presenting machinery in melanoma and other tumor cells in vitro and in vivo without increasing PD-L1 expression. Mechanistic studies indicated that 5-NL's induced MHC-I expression was inhibited by pharmacologically preventing cAMP Response Element-Binding Protein (CREB) phosphorylation. Importantly, 5-NL combined with anti-PD1 therapy showed significant improvement when compared to single anti-PD-1 treatment.
Conclusions: This study demonstrates novel therapeutic opportunities for augmenting immune responses in poorly immunogenic tumors.
Keywords: 5-Nonyloxytryptamine (5-NL); Antigen-presenting machinery; CD8+ T cells; Cold tumors; Immunotherapy; cAMP response element-binding protein (CREB).
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare no competing interests.
HCX, PP, KSL, and PAL declare that they are involved in the development of LCMV for clinical application in oncology in cooperation with or as advisors to Abalos Therapeutics GmbH. The other authors declare no competing interests.
Figures




References
-
- SEER Cancer Statistics Review, 1975–2014. 2017.
-
- Eggermont AM, Chiarion-Sileni V, Grob JJ, Dummer R, Wolchok JD, Schmidt H, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2015;16(5):522–530. doi: 10.1016/S1470-2045(15)70122-1. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

