Integrin β3 directly inhibits the Gα13-p115RhoGEF interaction to regulate G protein signaling and platelet exocytosis
- PMID: 37587112
- PMCID: PMC10432399
- DOI: 10.1038/s41467-023-40531-3
Integrin β3 directly inhibits the Gα13-p115RhoGEF interaction to regulate G protein signaling and platelet exocytosis
Abstract
The integrins and G protein-coupled receptors are both fundamental in cell biology. The cross talk between these two, however, is unclear. Here we show that β3 integrins negatively regulate G protein-coupled signaling by directly inhibiting the Gα13-p115RhoGEF interaction. Furthermore, whereas β3 deficiency or integrin antagonists inhibit integrin-dependent platelet aggregation and exocytosis (granule secretion), they enhance G protein-coupled RhoA activation and integrin-independent secretion. In contrast, a β3-derived Gα13-binding peptide or Gα13 knockout inhibits G protein-coupled RhoA activation and both integrin-independent and dependent platelet secretion without affecting primary platelet aggregation. In a mouse model of myocardial ischemia/reperfusion injury in vivo, the β3-derived Gα13-binding peptide inhibits platelet secretion of granule constituents, which exacerbates inflammation and ischemia/reperfusion injury. These data establish crucial integrin-G protein crosstalk, providing a rationale for therapeutic approaches that inhibit exocytosis in platelets and possibly other cells without adverse effects associated with loss of cell adhesion.
© 2023. Springer Nature Limited.
Conflict of interest statement
University of Illinois at Chicago holds patents related to this study. X.D. holds equity interests in DMT, Inc., which licenses UIC technology. The remaining authors declare no competing interests.
Figures
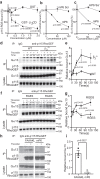
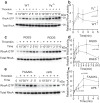
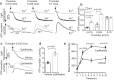
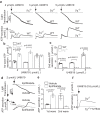

References
-
- Wootten D, Christopoulos A, Marti-Solano M, Babu MM, Sexton PM. Mechanisms of signalling and biased agonism in G protein-coupled receptors. Nat. Rev. Mol. Cell Biol. 2018;19:638–653. - PubMed
-
- Oldham WM, Hamm HE. Heterotrimeric G protein activation by G-protein-coupled receptors. Nat. Rev. Mol. Cell Biol. 2008;9:60–71. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

