Paraventricular Thalamic MC3R Circuits Link Energy Homeostasis with Anxiety-Related Behavior
- PMID: 37591737
- PMCID: PMC10490510
- DOI: 10.1523/JNEUROSCI.0704-23.2023
Paraventricular Thalamic MC3R Circuits Link Energy Homeostasis with Anxiety-Related Behavior
Abstract
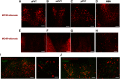
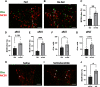
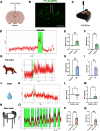
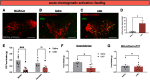
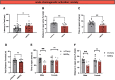
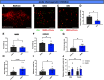
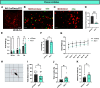
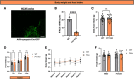
The hypothalamic melanocortin system is critically involved in sensing stored energy and communicating this information throughout the brain, including to brain regions controlling motivation and emotion. This system consists of first-order agouti-related peptide (AgRP) and pro-opiomelanocortin (POMC) neurons located in the hypothalamic arcuate nucleus and downstream neurons containing the melanocortin-3 (MC3R) and melanocortin-4 receptor (MC4R). Although extensive work has characterized the function of downstream MC4R neurons, the identity and function of MC3R-containing neurons are poorly understood. Here, we used neuroanatomical and circuit manipulation approaches in mice to identify a novel pathway linking hypothalamic melanocortin neurons to melanocortin-3 receptor neurons located in the paraventricular thalamus (PVT) in male and female mice. MC3R neurons in PVT are innervated by hypothalamic AgRP and POMC neurons and are activated by anorexigenic and aversive stimuli. Consistently, chemogenetic activation of PVT MC3R neurons increases anxiety-related behavior and reduces feeding in hungry mice, whereas inhibition of PVT MC3R neurons reduces anxiety-related behavior. These studies position PVT MC3R neurons as important cellular substrates linking energy status with neural circuitry regulating anxiety-related behavior and represent a promising potential target for diseases at the intersection of metabolism and anxiety-related behavior such as anorexia nervosa.SIGNIFICANCE STATEMENT Animals must constantly adapt their behavior to changing internal and external challenges, and impairments in appropriately responding to these challenges are a hallmark of many neuropsychiatric disorders. Here, we demonstrate that paraventricular thalamic neurons containing the melanocortin-3 receptor respond to energy-state-related information and external challenges to regulate anxiety-related behavior in mice. Thus, these neurons represent a potential target for understanding the neurobiology of disorders at the intersection of metabolism and psychiatry such as anorexia nervosa.
Keywords: MC3R; anxiety; feeding behavior; melanocortins; paraventricular thalamus.
Copyright © 2023 the authors.
Figures



Similar articles
-
Medial hypothalamic MC3R signalling regulates energy rheostasis in adult mice.J Physiol. 2025 Jan;603(2):379-410. doi: 10.1113/JP286699. Epub 2024 Dec 24. J Physiol. 2025. PMID: 39718394 Free PMC article.
-
Melanocortins and agouti-related protein modulate the excitability of two arcuate nucleus neuron populations by alteration of resting potassium conductances.J Physiol. 2007 Jan 15;578(Pt 2):425-38. doi: 10.1113/jphysiol.2006.119479. Epub 2006 Oct 26. J Physiol. 2007. PMID: 17068101 Free PMC article.
-
The melanocortin-3 receptor is a pharmacological target for the regulation of anorexia.Sci Transl Med. 2021 Apr 21;13(590):eabd6434. doi: 10.1126/scitranslmed.abd6434. Sci Transl Med. 2021. PMID: 33883274 Free PMC article.
-
POMC Neurons: Feeding, Energy Metabolism, and Beyond.Adv Exp Med Biol. 2018;1090:17-29. doi: 10.1007/978-981-13-1286-1_2. Adv Exp Med Biol. 2018. PMID: 30390283 Review.
-
The central melanocortin system and the integration of short- and long-term regulators of energy homeostasis.Recent Prog Horm Res. 2004;59:395-408. doi: 10.1210/rp.59.1.395. Recent Prog Horm Res. 2004. PMID: 14749511 Review.
Cited by
-
Fasting-induced activity changes in MC3R neurons of the paraventricular nucleus of the thalamus.Life Sci Alliance. 2024 Aug 6;7(10):e202402754. doi: 10.26508/lsa.202402754. Print 2024 Oct. Life Sci Alliance. 2024. PMID: 39107065 Free PMC article.
-
Neuronal activation in the paraventricular thalamus and lateral habenula during activity-based anorexia in mice.Neurosci Appl. 2024 Mar 6;3:104055. doi: 10.1016/j.nsa.2024.104055. eCollection 2024. Neurosci Appl. 2024. PMID: 40656074 Free PMC article.
-
Solitary and Synergistic Effects of Different Hydrophilic and Hydrophobic Phospholipid Moieties on Rat Behaviors.Pharmaceutics. 2024 Jun 4;16(6):762. doi: 10.3390/pharmaceutics16060762. Pharmaceutics. 2024. PMID: 38931883 Free PMC article.
-
Medial hypothalamic MC3R signalling regulates energy rheostasis in adult mice.J Physiol. 2025 Jan;603(2):379-410. doi: 10.1113/JP286699. Epub 2024 Dec 24. J Physiol. 2025. PMID: 39718394 Free PMC article.
-
Stress integration by an ascending adrenergic-melanocortin circuit.Neuropsychopharmacology. 2024 Aug;49(9):1361-1372. doi: 10.1038/s41386-024-01810-9. Epub 2024 Feb 7. Neuropsychopharmacology. 2024. PMID: 38326456 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
