Endothelial MICU1 alleviates diabetic cardiomyopathy by attenuating nitrative stress-mediated cardiac microvascular injury
- PMID: 37592255
- PMCID: PMC10436431
- DOI: 10.1186/s12933-023-01941-1
Endothelial MICU1 alleviates diabetic cardiomyopathy by attenuating nitrative stress-mediated cardiac microvascular injury
Abstract
Background: Myocardial microvascular injury is the key event in early diabetic heart disease. The injury of myocardial microvascular endothelial cells (CMECs) is the main cause and trigger of myocardial microvascular disease. Mitochondrial calcium homeostasis plays an important role in maintaining the normal function, survival and death of endothelial cells. Considering that mitochondrial calcium uptake 1 (MICU1) is a key molecule in mitochondrial calcium regulation, this study aimed to investigate the role of MICU1 in CMECs and explore its underlying mechanisms.
Methods: To examine the role of endothelial MICU1 in diabetic cardiomyopathy (DCM), we used endothelial-specific MICU1ecKO mice to establish a diabetic mouse model and evaluate the cardiac function. In addition, MICU1 overexpression was conducted by injecting adeno-associated virus 9 carrying MICU1 (AAV9-MICU1). Transcriptome sequencing technology was used to explore underlying molecular mechanisms.
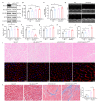
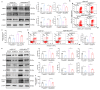
Results: Here, we found that MICU1 expression is decreased in CMECs of diabetic mice. Moreover, we demonstrated that endothelial cell MICU1 knockout exacerbated the levels of cardiac hypertrophy and interstitial myocardial fibrosis and led to a further reduction in left ventricular function in diabetic mice. Notably, we found that AAV9-MICU1 specifically upregulated the expression of MICU1 in CMECs of diabetic mice, which inhibited nitrification stress, inflammatory reaction, and apoptosis of the CMECs, ameliorated myocardial hypertrophy and fibrosis, and promoted cardiac function. Further mechanistic analysis suggested that MICU1 deficiency result in excessive mitochondrial calcium uptake and homeostasis imbalance which caused nitrification stress-induced endothelial damage and inflammation that disrupted myocardial microvascular endothelial barrier function and ultimately promoted DCM progression.
Conclusions: Our findings demonstrate that MICU1 expression was downregulated in the CMECs of diabetic mice. Overexpression of endothelial MICU1 reduced nitrification stress induced apoptosis and inflammation by inhibiting mitochondrial calcium uptake, which improved myocardial microvascular function and inhibited DCM progression. Our findings suggest that endothelial MICU1 is a molecular intervention target for the potential treatment of DCM.
Keywords: Cardiac microvascular endothelial cells (CMECs); Diabetic cardiomyopathy; Endothelial permeability; Inflammatory response; MICU1; Nitrative stress.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors state that they have no competing interests.
Figures






References
-
- Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, Colagiuri S, Guariguata L, Motala AA, Ogurtsova K, Shaw JE, Bright D, Williams R. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2019;157:107843. doi: 10.1016/j.diabres.2019.107843. - DOI - PubMed
-
- Joladarashi D, Zhu Y, Willman M, Nash K, Cimini M, Thandavarayan RA, Youker KA, Song X, Ren D, Li J, Kishore R, Krishnamurthy P, Wang L. STK35 gene therapy attenuates endothelial dysfunction and improves cardiac function in diabetes. Front Cardiovasc Med. 2021;8:798091. doi: 10.3389/fcvm.2021.798091. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

