Platelet factor XIII-A regulates platelet function and promotes clot retraction and stability
- PMID: 37601014
- PMCID: PMC10439398
- DOI: 10.1016/j.rpth.2023.100200
Platelet factor XIII-A regulates platelet function and promotes clot retraction and stability
Abstract
Background: Factor XIII (FXIII) is an important proenzyme in the hemostatic system. The plasma-derived enzyme activated FXIII cross-links fibrin fibers within thrombi to increase their mechanical strength and cross-links fibrin to fibrinolytic inhibitors, specifically α2-antiplasmin, to increase resistance to fibrinolysis. We have previously shown that cellular FXIII (factor XIII-A [FXIII-A]), which is abundant in the platelet cytoplasm, is externalized onto the activated membrane and cross-links extracellular substrates. The contribution of cellular FXIII-A to platelet activation and platelet function has not been extensively studied.
Objectives: This study aims to identify the role of platelet FXIII-A in platelet function.
Methods: We used normal healthy platelets with a cell permeable FXIII inhibitor and platelets from FXIII-deficient patients as a FXIII-free platelet model in a range of platelet function and clotting tests.
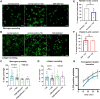
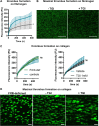
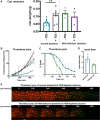
Results: Our data demonstrate that platelet FXIII-A enhances fibrinogen binding to the platelet surface upon agonist stimulation and improves the binding of platelets to fibrinogen and aggregation under flow in a whole-blood thrombus formation assay. In the absence of FXIII-A, platelets show reduced sensitivity to agonist stimulation, including decreased P-selectin exposure and fibrinogen binding. We show that FXIII-A is involved in platelet spreading where a lack of FXIII-A reduces the ability of platelets to fully spread on fibrinogen and collagen. Our data demonstrate that platelet FXIII-A is important for clot retraction where clots formed in its absence retracted to a lesser extent.
Conclusion: Overall, this study shows that platelet FXIII-A functions during thrombus formation by aiding platelet activation and thrombus retraction in addition to its antifibrinolytic roles.
Keywords: clot retraction; factor XIII; fibrinogen; fibrinolysis; platelet; platelet activation.
© 2023 The Authors.
Figures


References
-
- Gibbins J.M. Platelet adhesion signalling and the regulation of thrombus formation. J Cell Sci. 2004;117:3415–3425. - PubMed
-
- Whyte C.S., Mitchell J.L., Mutch N.J. Platelet-mediated modulation of fibrinolysis. Semin Thromb Hemost. 2017;43:115–128. - PubMed
-
- Schwartz M.L., Pizzo S.V., Hill R.L., McKee P.A. The subunit structures of human plasma and platelet factor XIII (fibrin-stabilizing factor) J Biol Chem. 1971;246:5851–5854. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

