Does the first generic exclusivity system provide an economic incentive for early generic entrance under the patent linkage system?
- PMID: 37601172
- PMCID: PMC10435867
- DOI: 10.3389/fpubh.2023.1120729
Does the first generic exclusivity system provide an economic incentive for early generic entrance under the patent linkage system?
Abstract
Introduction: A period of exclusivity for the first generics, as part of the patent linkage system, was established in South Korea to provide an economic incentive for early generic entry. This study describes the dynamics of generic penetration and assesses the first mover market share advantages under the patent linkage system.
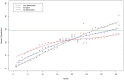
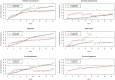
Methods: Pairs of originators and their corresponding generics granted the first generic exclusivity from 2015 to 2020 were identified. We categorized generics into first movers and latecomers, described the penetration curves of generics, and estimated the saturated market share of generics, first movers, and latecomers. Volume-based monthly prescriptions were used to describe the generics' penetration curves. A logistic growth model was adopted to estimate the saturated market shares of generics.
Results: We identified 28 pairs of originators and generics, presented penetration curves, and estimated generics market shares. The saturated market share of generics was 30%, and the time to saturation was approximately 33 months. The shapes of penetration varied by nationality, route, and number of generics. The existence of latecomers was associated with the decreased penetration speed over time and decreased market share of generics. However, the first mover market share advantages or latecomers' disadvantages were consistently observed.
Conclusion: The generic uptake in South Korea is delayed, limited, and context-dependent. However, first generics' market share advantage suggests that a period of exclusivity, as part of the patent linkage system, could provide an economic incentive for early generic entrance.
Keywords: South Korea; first generic exclusivity; incentive for early generic entry; patent linkage system; pharmaceutical market.
Copyright © 2023 Son.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Early-Stage Pharmaceutical Market Structures Between Originators and First Generics After Introducing the Patent Linkage System in South Korea.Int J Soc Determinants Health Health Serv. 2025 May 19:27551938251342990. doi: 10.1177/27551938251342990. Online ahead of print. Int J Soc Determinants Health Health Serv. 2025. PMID: 40388932
-
Market Exclusivity of the Originator Drugs in South Korea: A Retrospective Cohort Study.Front Public Health. 2021 Apr 6;9:654952. doi: 10.3389/fpubh.2021.654952. eCollection 2021. Front Public Health. 2021. PMID: 33889560 Free PMC article.
-
Generic atorvastatin and rosuvastatin in the South Korean market: time of introduction in relation to manufacturer characteristics.Expert Rev Pharmacoecon Outcomes Res. 2020 Oct;20(5):541-548. doi: 10.1080/14737167.2019.1664291. Epub 2019 Sep 10. Expert Rev Pharmacoecon Outcomes Res. 2020. PMID: 31483158
-
The Portuguese generic medicines market: What's next?Health Policy. 2020 Apr;124(4):397-403. doi: 10.1016/j.healthpol.2020.02.014. Epub 2020 Feb 29. Health Policy. 2020. PMID: 32139170 Review.
-
The Inflation Reduction Act: A boon for the generic and biosimilar industry.J Clin Pharm Ther. 2022 Nov;47(11):1738-1751. doi: 10.1111/jcpt.13783. Epub 2022 Oct 7. J Clin Pharm Ther. 2022. PMID: 36207987 Free PMC article. Review.
Cited by
-
Which factor reduces pharmaceutical expenditure, number of entrants or price variance? Updated generic drug markets in South Korea.Health Econ Rev. 2024 Aug 14;14(1):64. doi: 10.1186/s13561-024-00545-z. Health Econ Rev. 2024. PMID: 39141190 Free PMC article.
References
-
- Mirandah A. Patent linkage in Asian countries compared to the US. Asia IP Magazine. (2012) 4:50.
-
- Son K-B, Choi N, Lee B, Byun J, Yang D-W, Lee T-J. Patent challenges and factors associated with successful patent challengers under the patent linkage system: recent evidence from South Korea after the Korea United States free trade agreement. Glob Health. (2021) 17:1–10. doi: 10.1186/s12992-021-00765-6 - DOI - PMC - PubMed
-
- Correa CM. Bilateralism in intellectual property: defeating the WTO system for access to medicines. Case W Res J Int'l L. (2004) 36:79.
-
- Raju KD. Patent linkages and its impact on access to medicines: challenges, opportunities for developing countries. Access to Medicines and Vaccines, eds. Correa CM, Hilty RM. Springer. Gewerbestrasse: Springer. (2022).
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

