Early on-treatment C-reactive protein and its kinetics predict survival and response in recurrent and/or metastatic head and neck cancer patients receiving first-line pembrolizumab
- PMID: 37603206
- PMCID: PMC10560194
- DOI: 10.1007/s10637-023-01388-x
Early on-treatment C-reactive protein and its kinetics predict survival and response in recurrent and/or metastatic head and neck cancer patients receiving first-line pembrolizumab
Abstract
Purpose: First-line immune checkpoint blockade has improved the prognosis of recurrent and/or metastatic head and neck squamous cell carcinoma (R/M HNSCC), but response rates remain low. In this study, we aimed to investigate the prognostic value of CRP and its early kinetics to predict response and survival in R/M HNSCC.
Methods: A total of 87 patients who received first-line pembrolizumab for R/M HNSCC were analyzed. Three-fold cross-validation was used to estimate cut-off points of CRP at baseline and on-treatment (day 40 ± 10). Treatment response and survival were analyzed according to early CRP kinetics. The neutrophil-to-lymphocyte ratio (NLR) was used as a benchmark for the prognostic performance of CRP.
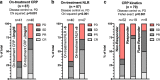
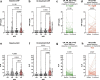
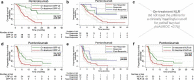
Results: On-treatment CRP below 2 mg/dl, 4x the upper limit of normal (ULN), was associated with increased overall survival (OS), while on-treatment CRP below 3 mg/dl (6x ULN) was correlated with a higher disease control rate (DCR) and increased progression-free survival (PFS). CRP flare-responders and CRP responders showed a higher DCR and longer PFS than CRP non-responders. An NLR above 6 was a negative prognosticator for progression. In multivariable analysis, on-treatment CRP prevailed as the only significant prognosticator for OS (HR: 4.97, CI95%: 2.18-11.32, p < 0.001) and PFS (HR: 2.07, CI95%: 1.07-3.99, p = 0.030).
Conclusion: On-treatment CRP was identified as a prognostic biomarker for objective response and survival in R/M HNSCC patients receiving first-line pembrolizumab and could be easily incorporated into clinical practice as a widely available and cost-effective biomarker.
Keywords: Biomarkers; C-Reactive protein; Head and Neck Neoplasms; Immunotherapy; Kinetics.
© 2023. The Author(s).
Conflict of interest statement
Thorsten Fuereder received honoraria from MSD, Merck Darmstadt, Roche, BMS, Accord, Sanofi, Boehringer Ingelheim, Amgen, Pfizer and Takeda, and research grants from Merck and MSD. The other authors declare no conflict of interest related to the publication of this article.
Figures
References
-
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the global burden of Disease Study 2016. Lancet. 2017;390:1211–1259. doi: 10.1016/s0140-6736(17)32154-2. - DOI - PMC - PubMed
-
- Burtness B, et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet. 2019;394:1915–1928. doi: 10.1016/s0140-6736(19)32591-7. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

