Epigenetic histone modification by butyrate downregulates KIT and attenuates mast cell function
- PMID: 37603611
- PMCID: PMC10538265
- DOI: 10.1111/jcmm.17924
Epigenetic histone modification by butyrate downregulates KIT and attenuates mast cell function
Abstract
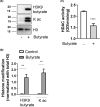
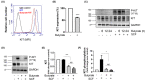
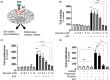
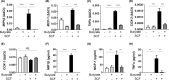
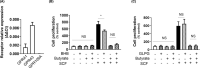
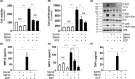
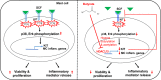
Short-chain fatty acid butyrate is produced from the bacterial fermentation of indigestible fiber in the intestinal lumen, and it has been shown to attenuate lung inflammation in murine asthma models. Mast cells (MCs) are initiators of inflammatory response to allergens, and they play an important role in asthma. MC survival and proliferation is regulated by its growth factor stem cell factor (SCF), which acts through the receptor, KIT. It has previously been shown that butyrate attenuates the activation of MCs by allergen stimulation. However, how butyrate mechanistically influences SCF signalling to impact MC function remains unknown. Here, we report that butyrate treatment triggered the modification of MC histones via butyrylation and acetylation, and inhibition of histone deacetylase (HDAC) activity. Further, butyrate treatment caused downregulation of SCF receptor KIT and associated phosphorylation, leading to significant attenuation of SCF-mediated MC proliferation, and pro-inflammatory cytokine secretion. Mechanistically, butyrate inhibited MC function by suppressing KIT and downstream p38 and Erk phosphorylation, and it mediated these effects via modification of histones, acting as an HDAC inhibitor and not via its traditional GPR41 (FFAR3) or GPR43 (FFAR2) butyrate receptors. In agreement, the pharmacological inhibition of Class I HDAC (HDAC1/3) mirrored butyrate's effects, suggesting that butyrate impacts MC function by HDAC1/3 inhibition. Taken together, butyrate epigenetically modifies histones and downregulates the SCF/KIT/p38/Erk signalling axis, leading to the attenuation of MC function, validating its ability to suppress MC-mediated inflammation. Therefore, butyrate supplementations could offer a potential treatment strategy for allergy and asthma via epigenetic alterations in MCs.
Keywords: HDAC; KIT; MAPK; MC; SCF; asthma; butyrate; proliferation; viability.
© 2023 The Authors. Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
Authors declare no conflict of interest.
Figures

References
-
- Gurish MF, Boyce JA. Mast cells: ontogeny, homing, and recruitment of a unique innate effector cell. J Allergy Clin Immunol. 2006;117(6):1285‐1291. - PubMed
-
- Galli SJ, Kalesnikoff J, Grimbaldeston MA, Piliponsky AM, Williams CM, Tsai M. Mast cells as "tunable" effector and immunoregulatory cells: recent advances. Annu Rev Immunol. 2005;23:749‐786. - PubMed
-
- Galli SJ, Tsai M, Wershil BK, Tam SY, Costa JJ. Regulation of mouse and human mast cell development, survival and function by stem cell factor, the ligand for the c‐kit receptor. Int Arch Allergy Immunol. 1995;107(1–3):51‐53. - PubMed
-
- Metcalfe DD, Baram D, Mekori YA. Mast cells. Physiol Rev. 1997;77(4):1033‐1079. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

