A new Hoxb8FlpO mouse line for intersectional approaches to dissect developmentally defined adult sensorimotor circuits
- PMID: 37603775
- PMCID: PMC10437123
- DOI: 10.3389/fnmol.2023.1176823
A new Hoxb8FlpO mouse line for intersectional approaches to dissect developmentally defined adult sensorimotor circuits
Abstract
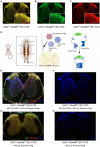
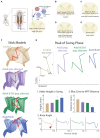
Improvements in the speed and cost of expression profiling of neuronal tissues offer an unprecedented opportunity to define ever finer subgroups of neurons for functional studies. In the spinal cord, single cell RNA sequencing studies support decades of work on spinal cord lineage studies, offering a unique opportunity to probe adult function based on developmental lineage. While Cre/Flp recombinase intersectional strategies remain a powerful tool to manipulate spinal neurons, the field lacks genetic tools and strategies to restrict manipulations to the adult mouse spinal cord at the speed at which new tools develop. This study establishes a new workflow for intersectional mouse-viral strategies to dissect adult spinal function based on developmental lineages in a modular fashion. To restrict manipulations to the spinal cord, we generate a brain-sparing Hoxb8FlpO mouse line restricting Flp recombinase expression to caudal tissue. Recapitulating endogenous Hoxb8 gene expression, Flp-dependent reporter expression is present in the caudal embryo starting day 9.5. This expression restricts Flp activity in the adult to the caudal brainstem and below. Hoxb8FlpO heterozygous and homozygous mice do not develop any of the sensory or locomotor phenotypes evident in Hoxb8 heterozygous or mutant animals, suggesting normal developmental function of the Hoxb8 gene and protein in Hoxb8FlpO mice. Compared to the variability of brain recombination in available caudal Cre and Flp lines, Hoxb8FlpO activity is not present in the brain above the caudal brainstem, independent of mouse genetic background. Lastly, we combine the Hoxb8FlpO mouse line with dorsal horn developmental lineage Cre mouse lines to express GFP in developmentally determined dorsal horn populations. Using GFP-dependent Cre recombinase viruses and Cre recombinase-dependent inhibitory chemogenetics, we target developmentally defined lineages in the adult. We show how developmental knock-out versus transient adult silencing of the same ROR𝛃 lineage neurons affects adult sensorimotor behavior. In summary, this new mouse line and viral approach provides a blueprint to dissect adult somatosensory circuit function using Cre/Flp genetic tools to target spinal cord interneurons based on genetic lineage.
Keywords: dorsal root ganglion (DRG); genetic tools; neuroscience; sensorimotor system; spinal cord.
Copyright © 2023 Bohic, Upadhyay, Eisdorfer, Keating, Simon, Briones, Azadegan, Nacht, Oputa, Martinez, Bethell, Gradwell, Romanienko, Ramer, Stuber and Abraira.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Direct comparison of Hoxb8-driven reporter distribution in the brains of four transgenic mouse lines: towards a spinofugal projection atlas.Front Neuroanat. 2024 May 16;18:1400015. doi: 10.3389/fnana.2024.1400015. eCollection 2024. Front Neuroanat. 2024. PMID: 38817241 Free PMC article.
-
Differential CRH expression level determines efficiency of Cre- and Flp-dependent recombination.Front Neurosci. 2023 Aug 3;17:1163462. doi: 10.3389/fnins.2023.1163462. eCollection 2023. Front Neurosci. 2023. PMID: 37599997 Free PMC article.
-
tTARGIT AAVs mediate the sensitive and flexible manipulation of intersectional neuronal populations in mice.Elife. 2021 Mar 11;10:e66835. doi: 10.7554/eLife.66835. Elife. 2021. PMID: 33704065 Free PMC article.
-
A Phox2b::FLPo transgenic mouse line suitable for intersectional genetics.Genesis. 2013 Jul;51(7):506-14. doi: 10.1002/dvg.22393. Epub 2013 May 22. Genesis. 2013. PMID: 23592597 Free PMC article.
-
Flp-recombinase mouse line for genetic manipulation of ipRGCs.bioRxiv [Preprint]. 2024 May 8:2024.05.06.592761. doi: 10.1101/2024.05.06.592761. bioRxiv. 2024. PMID: 38766000 Free PMC article. Preprint.
Cited by
-
Direct comparison of Hoxb8-driven reporter distribution in the brains of four transgenic mouse lines: towards a spinofugal projection atlas.Front Neuroanat. 2024 May 16;18:1400015. doi: 10.3389/fnana.2024.1400015. eCollection 2024. Front Neuroanat. 2024. PMID: 38817241 Free PMC article.
-
Keratinocyte-TRPV1 sensory neuron interactions in a genetically controllable mouse model of chronic neuropathic itch.Proc Natl Acad Sci U S A. 2025 Jun 17;122(24):e2411724122. doi: 10.1073/pnas.2411724122. Epub 2025 Jun 11. Proc Natl Acad Sci U S A. 2025. PMID: 40498457 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

