Phytochemical mediated modulation of COX-3 and NFκB for the management and treatment of arthritis
- PMID: 37604838
- PMCID: PMC10442333
- DOI: 10.1038/s41598-023-37729-2
Phytochemical mediated modulation of COX-3 and NFκB for the management and treatment of arthritis
Abstract
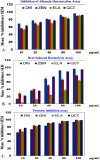
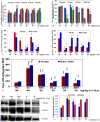
In this study, we investigated whether zerumbone (ZBN), ellagic acid (ELA) and quercetin (QCT), the plant-derived components, can modulate the role of COX-3 or cytokines liable in arthritic disorder. Initially, the effect of ZBN, ELA, and QCT on inflammatory process was investigated using in-vitro models. In-silico docking and molecular dynamics study of these molecules with respective targets also corroborate with in-vitro studies. Further, the in-vivo anti-arthritic potential of these molecules in Complete Freund's adjuvant (CFA)-induced arthritic rats was confirmed. CFA increases in TNF-α and IL-1β levels in the arthritic control animals were significantly (***p < 0.001) attenuated in the ZBN- and ELA-treated animals. CFA-induced attenuation in IL-10 levels recovered under treatment. Moreover, ELA attenuated CFA-induced upregulation of COX-3 and ZBN downregulated CFA-triggered NFκB expression in arthritic animals. The bonding patterns of zerumbone in the catalytic sites of targets provide a useful hint in designing and developing suitable derivatives that can be used as a potential drug. To our best knowledge, the first time we are reporting the role of COX-3 in the treatment of arthritic disorders which could provide a novel therapeutic approach for the treatment of inflammatory disorders.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures

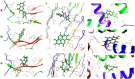

Similar articles
-
Ephedra gerardiana aqueous ethanolic extract and fractions attenuate Freund Complete Adjuvant induced arthritis in Sprague Dawley rats by downregulating PGE2, COX2, IL-1β, IL-6, TNF-α, NF-kB and upregulating IL-4 and IL-10.J Ethnopharmacol. 2018 Oct 5;224:482-496. doi: 10.1016/j.jep.2018.06.018. Epub 2018 Jun 18. J Ethnopharmacol. 2018. PMID: 29920358
-
Novel anti-arthritic mechanisms of trans-cinnamaldehyde against complete Freund's adjuvant-induced arthritis in mice: involvement of NF-кB/TNF-α and IL-6/IL-23/ IL-17 pathways in the immuno-inflammatory responses.Inflammopharmacology. 2022 Oct;30(5):1769-1780. doi: 10.1007/s10787-022-01005-y. Epub 2022 Jun 1. Inflammopharmacology. 2022. PMID: 35648328 Free PMC article.
-
Nerolidol: a potential approach in rheumatoid arthritis through reduction of TNF-α, IL-1β, IL-6, NF-kB, COX-2 and antioxidant effect in CFA-induced arthritic model.Inflammopharmacology. 2022 Apr;30(2):537-548. doi: 10.1007/s10787-022-00930-2. Epub 2022 Feb 25. Inflammopharmacology. 2022. PMID: 35212850
-
Anti-arthritic activity of ferulic acid in complete Freund's adjuvant (CFA)-induced arthritis in rats: JAK2 inhibition.Inflammopharmacology. 2020 Apr;28(2):463-473. doi: 10.1007/s10787-019-00642-0. Epub 2019 Sep 27. Inflammopharmacology. 2020. PMID: 31562605
-
Nimbolide exerts protective effects in complete Freund's adjuvant induced inflammatory arthritis via abrogation of STAT-3/NF-κB/Notch-1 signaling.Life Sci. 2021 Feb 1;266:118911. doi: 10.1016/j.lfs.2020.118911. Epub 2020 Dec 16. Life Sci. 2021. PMID: 33333049
Cited by
-
Paracetamol: the potential therapeutic pathways defining its clinical use.Inflammopharmacology. 2025 Jun;33(6):2907-2918. doi: 10.1007/s10787-025-01779-x. Epub 2025 May 22. Inflammopharmacology. 2025. PMID: 40402381 Review.
-
Novel antiarthritic mechanisms of Azelaic acid against CFA-induced arthritis in rats by modulating pro- and anti-inflammatory cytokines network.Inflammopharmacology. 2024 Aug;32(4):2445-2462. doi: 10.1007/s10787-024-01512-0. Epub 2024 Jun 25. Inflammopharmacology. 2024. PMID: 38916711
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

