Allogeneic Hematopoietic Cell Transplantation Improves Outcome in Myelodysplastic Syndrome Across High-Risk Genetic Subgroups: Genetic Analysis of the Blood and Marrow Transplant Clinical Trials Network 1102 Study
- PMID: 37607457
- PMCID: PMC10552956
- DOI: 10.1200/JCO.23.00866
Allogeneic Hematopoietic Cell Transplantation Improves Outcome in Myelodysplastic Syndrome Across High-Risk Genetic Subgroups: Genetic Analysis of the Blood and Marrow Transplant Clinical Trials Network 1102 Study
Abstract
Purpose: Allogeneic hematopoietic cell transplantation (HCT) in patients with myelodysplastic syndrome (MDS) improves overall survival (OS). We evaluated the impact of MDS genetics on the benefit of HCT in a biological assignment (donor v no donor) study.
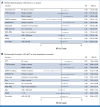
Methods: We performed targeted sequencing in 309 patients age 50-75 years with International Prognostic Scoring System (IPSS) intermediate-2 or high-risk MDS, enrolled in the Blood and Marrow Transplant Clinical Trials Network 1102 study and assessed the association of gene mutations with OS. Patients with TP53 mutations were classified as TP53multihit if two alleles were altered (via point mutation, deletion, or copy-neutral loss of heterozygosity).
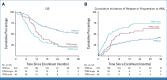
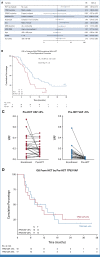
Results: The distribution of gene mutations was similar in the donor and no donor arms, with TP53 (28% v 29%; P = .89), ASXL1 (23% v 29%; P = .37), and SRSF2 (16% v 16%; P = .99) being most common. OS in patients with a TP53 mutation was worse compared with patients without TP53 mutation (21% ± 5% [SE] v 52% ± 4% at 3 years; P < .001). Among those with a TP53 mutation, OS was similar between TP53single versus TP53multihit (22% ± 8% v 20% ± 6% at 3 years; P = .31). Considering HCT as a time-dependent covariate, patients with a TP53 mutation who underwent HCT had improved OS compared with non-HCT treatment (OS at 3 years: 23% ± 7% v 11% ± 7%; P = .04), associated with a hazard ratio of 3.89; 95% CI, 1.87 to 8.12; P < .001 after adjustment for covariates. OS among patients with molecular IPSS (IPSS-M) very high risk without a TP53 mutation was significantly improved if they had a donor (68% ± 10% v 0% ± 12% at 3 years; P = .001).
Conclusion: HCT improved OS compared with non-HCT treatment in patients with TP53 mutations irrespective of TP53 allelic status. Patients with IPSS-M very high risk without a TP53 mutation had favorable outcomes when a donor was available.
Trial registration: ClinicalTrials.gov NCT02016781.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
This author is a member of the
No other potential conflicts of interest were reported.
Figures


References
-
- Kröger N, Sockel K, Wolschke C, et al. : Comparison between 5-azacytidine treatment and allogeneic stem-cell transplantation in elderly patients with advanced MDS according to donor availability (VidazaAllo study). J Clin Oncol 39:3318-3327, 2021 - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

