The current state of research on influenza antiviral drug development: drugs in clinical trial and licensed drugs
- PMID: 37610204
- PMCID: PMC10653855
- DOI: 10.1128/mbio.01273-23
The current state of research on influenza antiviral drug development: drugs in clinical trial and licensed drugs
Retraction in
-
Retraction for Li et al., "The current state of research on influenza antiviral drug development: drugs in clinical trial and licensed drugs".mBio. 2024 Apr 10;15(4):e0010624. doi: 10.1128/mbio.00106-24. Epub 2024 Mar 22. mBio. 2024. PMID: 38517163 Free PMC article. No abstract available.
Corrected and republished in
-
Retracted and republished from: "The current state of research on influenza antiviral drug development: drugs in clinical trial and licensed drugs".mBio. 2024 May 8;15(5):e0017524. doi: 10.1128/mbio.00175-24. Epub 2024 Mar 29. mBio. 2024. PMID: 38551343 Free PMC article.
Abstract
Influenza viruses (IVs) threaten global human health due to the high morbidity, infection, and mortality rates. Currently, the influenza drugs recommended by the FDA are oseltamivir, zanamivir, peramivir, and baloxavir marboxil. Notably, owing to the high variability of IVs, no drug exists that can effectively treat all types and subtypes of IVs. Moreover, the current trend of drug resistance is likely to continue as the viral genome is constantly mutating. Therefore, there is an urgent need to develop drugs related to the treatment of influenza to deal with the next pandemic. Here, we summarized the cutting-edge research in mechanism of action, inhibitory activity, and clinical efficacy of drugs that have been approved and drugs that are still in clinical trials for influenza treatment. We hope this review will provide up-to-date and comprehensive information on influenza antivirals and generate hypotheses for screens and development of new broad-spectrum influenza drugs in the near future.
Keywords: adverse event; antiviral drugs; clinical drugs; influenza virus; mechanism of action.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
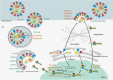

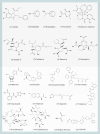

References
-
- WHO . 2023. Influenza (seasonal). Available from: https://www.who.int/health-topics/influenza-seasonal
-
- CDC . 2023. Specific avian flu viruses. Available from: https://www.cdc.gov/flu/avianflu/specific-flu-viruses.htm
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

