In Vivo Electron Paramagnetic Resonance Molecular Profiling of Tumor Microenvironment upon Tumor Progression to Malignancy in an Animal Model of Breast Cancer
- PMID: 37610610
- PMCID: PMC10884355
- DOI: 10.1007/s11307-023-01847-0
In Vivo Electron Paramagnetic Resonance Molecular Profiling of Tumor Microenvironment upon Tumor Progression to Malignancy in an Animal Model of Breast Cancer
Abstract
Purpose: Hypoxia and acidosis are recognized tumor microenvironment (TME) biomarkers of cancer progression. Alterations in cancer redox status and metabolism are also associated with elevated levels of intracellular glutathione (GSH) and interstitial inorganic phosphate (Pi). This study aims to evaluate the capability of these biomarkers to discriminate between stages and inform on a switch to malignancy.
Procedures: These studies were performed using MMTV-PyMT( +) female transgenic mice that spontaneously develop breast cancer and emulate human tumor staging. In vivo assessment of oxygen concentration (pO2), extracellular acidity (pHe), Pi, and GSH was performed using L-band electron paramagnetic resonance spectroscopy and multifunctional trityl and GSH-sensitive nitroxide probes.
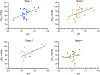
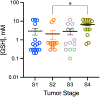
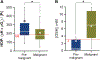
Results: Profiling of the TME showed significant deviation of measured biomarkers upon tumor progression from pre-malignancy (pre-S4) to the malignant stage (S4). For the combined marker, HOP: (pHe × pO2)/Pi, a value > 186 indicated that the tumors were pre-malignant in 85% of the mammary glands analyzed, and when < 186, they were malignant 42% of the time. For GSH, a value < 3 mM indicated that the tumors were pre-malignant 74% of the time, and when > 3 mM, they were malignant 80% of the time. The only marker that markedly deviated as early as stage 1 (S1) from its value in pre-S1 was elevated Pi, followed by a decrease of pHe and pO2 and increase in GSH at later stages.
Conclusion: Molecular TME profiling informs on alteration of tumor redox and metabolism during tumor staging. Early elevation of interstitial Pi at S1 may reflect tumor metabolic alterations that demand elevated phosphorus supply in accordance with the high rate growth hypothesis. These metabolic changes are supported by the following decrease of pHe due to a high tumor reliance on glycolysis and increase of intracellular GSH, a major intracellular redox buffer. The appreciable decrease in TME pO2 was observed only at malignant S4, apparently as a consequence of tumor mass growth and corresponding decrease in perfusion efficacy and increase in oxygen consumption as the tumor cells proliferate.
Keywords: Electron paramagnetic resonance; Interstitial inorganic phosphate; Intracellular glutathione; Molecular cancer biomarkers; Nitroxides; Spin probes; Trityl radicals; Tumor hypoxia and acidosis; Tumor microenvironment.
© 2023. The Author(s), under exclusive licence to World Molecular Imaging Society.
Conflict of interest statement
Figures
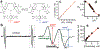
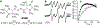
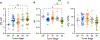
Similar articles
-
Interstitial Inorganic Phosphate as a Tumor Microenvironment Marker for Tumor Progression.Sci Rep. 2017 Jan 24;7:41233. doi: 10.1038/srep41233. Sci Rep. 2017. PMID: 28117423 Free PMC article.
-
In Vivo Molecular Electron Paramagnetic Resonance-Based Spectroscopy and Imaging of Tumor Microenvironment and Redox Using Functional Paramagnetic Probes.Antioxid Redox Signal. 2018 May 20;28(15):1365-1377. doi: 10.1089/ars.2017.7329. Epub 2017 Dec 20. Antioxid Redox Signal. 2018. PMID: 29132215 Free PMC article. Review.
-
In Vivo EPR Assessment of pH, pO2, Redox Status, and Concentrations of Phosphate and Glutathione in the Tumor Microenvironment.J Vis Exp. 2018 Mar 16;(133):56624. doi: 10.3791/56624. J Vis Exp. 2018. PMID: 29608148 Free PMC article.
-
Biocompatible Monophosphonated Trityl Spin Probe, HOPE71, for In Vivo Measurement of pO2, pH, and [Pi] by Electron Paramagnetic Resonance Spectroscopy.Anal Chem. 2023 Jan 17;95(2):946-954. doi: 10.1021/acs.analchem.2c03476. Epub 2022 Dec 20. Anal Chem. 2023. PMID: 36537829 Free PMC article.
-
Janus-faced tumor microenvironment and redox.Antioxid Redox Signal. 2014 Aug 10;21(5):723-9. doi: 10.1089/ars.2014.5864. Epub 2014 Mar 4. Antioxid Redox Signal. 2014. PMID: 24512276 Free PMC article. Review.
Cited by
-
Sensitive simultaneous measurements of oxygenation and extracellular pH by EPR using a stable monophosphonated trityl radical and lithium phthalocyanine.Free Radic Biol Med. 2024 Mar;213:11-18. doi: 10.1016/j.freeradbiomed.2024.01.012. Epub 2024 Jan 11. Free Radic Biol Med. 2024. PMID: 38218552 Free PMC article.
-
Line-Shifting Triarylmethyl Radicals for Imaging of Enzyme Activity Using Overhauser-Enhanced Magnetic Resonance Imaging: Application to Alkaline Phosphatase.Chem Biomed Imaging. 2024 Nov 23;3(1):35-44. doi: 10.1021/cbmi.4c00059. eCollection 2025 Jan 27. Chem Biomed Imaging. 2024. PMID: 39886221 Free PMC article.
-
Carcinogenesis Associated with Toxin Nephropathy: Proposed Mediation by Phosphate Toxicity.Cells. 2025 Jun 22;14(13):952. doi: 10.3390/cells14130952. Cells. 2025. PMID: 40643473 Free PMC article. Review.
-
Assessing the Therapeutic Efficacy of Proton Transport Inhibitors in a Triple-Negative Breast Cancer Murine Model with Magnetic Resonance Imaging-Chemical Exchange Saturation Transfer Tumor pH Imaging.Metabolites. 2023 Nov 18;13(11):1161. doi: 10.3390/metabo13111161. Metabolites. 2023. PMID: 37999256 Free PMC article.
-
Spontaneous Tumor Regression and Reversion: Insights and Associations with Reduced Dietary Phosphate.Cancers (Basel). 2024 Jun 3;16(11):2126. doi: 10.3390/cancers16112126. Cancers (Basel). 2024. PMID: 38893245 Free PMC article. Review.
References
-
- Siemann DW, ed. Tumor Microenvironment. 2011, John Wiley & Sons, Ltd.: Chichester, UK; Hoboken, NJ, USA. 436.
-
- Tatum JL, Kelloff GJ, Gillies RJ et al. (2006) Hypoxia: importance in tumor biology, noninvasive measurement by imaging, and value of its measurement in the management of cancer therapy. Int J Radiat Biol 82(10): 699–757. - PubMed
-
- Brahimi-Horn MC, Chiche J, Pouyssegur J (2007) Hypoxia signalling controls metabolic demand. Curr Opin Cell Biol 19(2): 223–229. - PubMed
-
- Haulica A, Ababei L (1974) Comparative study of glycolytic activity in the erythrocytes of animals with chronic experimental hypoxia and with tumours. Neoplasma 21(1): 29–35. - PubMed
MeSH terms
Substances
Grants and funding
- R01 CA194013/CA/NCI NIH HHS/United States
- R00 EB023990/EB/NIBIB NIH HHS/United States
- CA194013/CA/NCI NIH HHS/United States
- K99 EB023990/EB/NIBIB NIH HHS/United States
- U54 GM104942/GM/NIGMS NIH HHS/United States
- R01 CA192064/CA/NCI NIH HHS/United States
- U54GM104942/GM/NIGMS NIH HHS/United States
- EB023990/EB/NIBIB NIH HHS/United States
- CA192064/CA/NCI NIH HHS/United States
- EB028553/EB/NIBIB NIH HHS/United States
- R21 EB028553/EB/NIBIB NIH HHS/United States
- CA192064/CA/NCI NIH HHS/United States
- CA194013/CA/NCI NIH HHS/United States
- EB028553/EB/NIBIB NIH HHS/United States
- EB023990/EB/NIBIB NIH HHS/United States
- U54GM104942/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

