Prenatal Exposure to Maternal Mood Entropy Is Associated With a Weakened and Inflexible Salience Network in Adolescence
- PMID: 37611745
- PMCID: PMC10881896
- DOI: 10.1016/j.bpsc.2023.08.002
Prenatal Exposure to Maternal Mood Entropy Is Associated With a Weakened and Inflexible Salience Network in Adolescence
Abstract
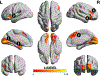
Background: Fetal exposure to maternal mood dysregulation influences child cognitive and emotional development, which may have long-lasting implications for mental health. However, the neurobiological alterations associated with this dimension of adversity have yet to be explored. Here, we tested the hypothesis that fetal exposure to entropy, a novel index of dysregulated maternal mood, would predict the integrity of the salience network, which is involved in emotional processing.
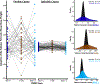
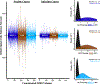
Methods: A sample of 138 child-mother pairs (70 females) participated in this prospective longitudinal study. Maternal negative mood level and entropy (an index of variable and unpredictable mood) were assessed 5 times during pregnancy. Adolescents engaged in a functional magnetic resonance imaging task that was acquired between 2 resting-state scans. Changes in network integrity were analyzed using mixed-effect and latent growth curve models. The amplitude of low frequency fluctuations was analyzed to corroborate findings.
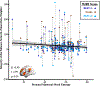
Results: Prenatal maternal mood entropy, but not mood level, was associated with salience network integrity. Both prenatal negative mood level and entropy were associated with the amplitude of low frequency fluctuations of the salience network. Latent class analysis yielded 2 profiles based on changes in network integrity across all functional magnetic resonance imaging sequences. The profile that exhibited little variation in network connectivity (i.e., inflexibility) consisted of adolescents who were exposed to higher negative maternal mood levels and more entropy.
Conclusions: These findings suggest that fetal exposure to maternal mood dysregulation is associated with a weakened and inflexible salience network. More broadly, they identify maternal mood entropy as a novel marker of early adversity that exhibits long-lasting associations with offspring brain development.
Keywords: Dysregulation; Emotional processing; Maternal mood entropy; Neurodevelopment; Salience network; fMRI.
Copyright © 2023 Society of Biological Psychiatry. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
DISCLOSURES
The authors report no biomedical financial interests or potential conflicts of interest.
Figures
Similar articles
-
Prenatal maternal mood patterns predict child temperament and adolescent mental health.J Affect Disord. 2018 Mar 1;228:83-90. doi: 10.1016/j.jad.2017.11.065. Epub 2017 Nov 14. J Affect Disord. 2018. PMID: 29241049 Free PMC article.
-
Maternal Immune Activation During the Third Trimester Is Associated with Neonatal Functional Connectivity of the Salience Network and Fetal to Toddler Behavior.J Neurosci. 2018 Mar 14;38(11):2877-2886. doi: 10.1523/JNEUROSCI.2272-17.2018. Epub 2018 Feb 27. J Neurosci. 2018. PMID: 29487127 Free PMC article.
-
Prenatal maternal mood entropy is associated with child neurodevelopment.Emotion. 2021 Apr;21(3):489-498. doi: 10.1037/emo0000726. Epub 2020 Mar 23. Emotion. 2021. PMID: 32202848 Free PMC article.
-
Prenatal exposure to air pollution and maternal depression: Combined effects on brain aging and mental health in young adulthood.Prog Neuropsychopharmacol Biol Psychiatry. 2024 Aug 30;134:111062. doi: 10.1016/j.pnpbp.2024.111062. Epub 2024 Jun 19. Prog Neuropsychopharmacol Biol Psychiatry. 2024. PMID: 38906411
-
Longitudinal changes in within-salience network functional connectivity mediate the relationship between childhood abuse and neglect, and mental health during adolescence.Psychol Med. 2023 Mar;53(4):1552-1564. doi: 10.1017/S0033291721003135. Epub 2021 Aug 25. Psychol Med. 2023. PMID: 34429171
Cited by
-
Individual longitudinal changes in DNA-methylome identify signatures of early-life adversity and correlate with later outcome.Neurobiol Stress. 2024 May 31;31:100652. doi: 10.1016/j.ynstr.2024.100652. eCollection 2024 Jul. Neurobiol Stress. 2024. PMID: 38962694 Free PMC article.
-
Exposure to unpredictable childhood environments is associated with amygdala activation during early extinction in adulthood.Dev Cogn Neurosci. 2025 Aug;74:101578. doi: 10.1016/j.dcn.2025.101578. Epub 2025 May 31. Dev Cogn Neurosci. 2025. PMID: 40493990 Free PMC article.
-
Within-subject changes in methylome profile identify individual signatures of early-life adversity, with a potential to predict neuropsychiatric outcome.bioRxiv [Preprint]. 2023 Dec 19:2023.12.16.571594. doi: 10.1101/2023.12.16.571594. bioRxiv. 2023. Update in: Neurobiol Stress. 2024 May 31;31:100652. doi: 10.1016/j.ynstr.2024.100652. PMID: 38187766 Free PMC article. Updated. Preprint.
-
Across ages and places: Unpredictability of maternal sensory signals and child internalizing behaviors.J Affect Disord. 2024 Feb 15;347:557-567. doi: 10.1016/j.jad.2023.11.068. Epub 2023 Nov 23. J Affect Disord. 2024. PMID: 38007106 Free PMC article.
-
Childhood unpredictability is associated with increased risk for long- and short-term depression and anhedonia symptoms following combat deployment.J Mood Anxiety Disord. 2024 Jun;6:100045. doi: 10.1016/j.xjmad.2023.100045. Epub 2023 Dec 23. J Mood Anxiety Disord. 2024. PMID: 38911511 Free PMC article.
References
-
- Sandman CA, Class QA, Glynn LM, Davis EP (2016): Chapter 13-Neurobehavioral disorders and developmental origins of health and disease. In: Rosenfeld CS, editor. The Epigenome and Developmental Origins of Health and Disease. Boston: Academic Press, 235–266.

