Sustained delivery of osteogenic growth peptide through injectable photoinitiated composite hydrogel for osteogenesis
- PMID: 37614629
- PMCID: PMC10444198
- DOI: 10.3389/fbioe.2023.1228250
Sustained delivery of osteogenic growth peptide through injectable photoinitiated composite hydrogel for osteogenesis
Abstract
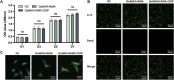
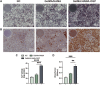
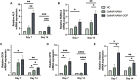
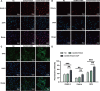
One of the most challenging clinical issues continues to be the effective bone regeneration and rebuilding following bone abnormalities. Although osteogenic growth peptide (OGP) has been proven to be effective in promoting osteoblast activity, its clinical application is constrained by abrupt release and easily degradation. We developed a GelMA/HAMA dual network hydrogel loaded with OGP based on a combination of physical chain entanglement and chemical cross-linking effects to produce an efficient long-term sustained release of OGP. The hydrogel polymers were quickly molded under ultraviolet (UV) light and had the suitable physical characteristics, porosity structure and biocompatibility. Significantly, the GelMA/HAMA-OGP hydrogel could promote cell proliferation, adhesion, increase osteogenic-related gene and protein expression in vitro. In conclusion, the OGP sustained-release system based on GelMA/HAMA dual network hydrogel offers a fresh perspective on bone regeneration therapy.
Keywords: GelMA; HAMA; OGP; bone tissue engineering; photo-crosslinking; sustained release.
Copyright © 2023 Liu, Wu, Sun, Meng and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Gelatin Templated Polypeptide Co-Cross-Linked Hydrogel for Bone Regeneration.Adv Healthc Mater. 2020 Jan;9(1):e1901239. doi: 10.1002/adhm.201901239. Epub 2019 Dec 9. Adv Healthc Mater. 2020. PMID: 31814318
-
Osteogenic growth peptide (OGP)-loaded amphiphilic peptide (NapFFY) supramolecular hydrogel promotes osteogenesis and bone tissue reconstruction.Int J Biol Macromol. 2022 Jan 15;195:558-564. doi: 10.1016/j.ijbiomac.2021.12.028. Epub 2021 Dec 14. Int J Biol Macromol. 2022. PMID: 34920074
-
Human urine-derived stem cell exosomes delivered via injectable GelMA templated hydrogel accelerate bone regeneration.Mater Today Bio. 2023 Feb 1;19:100569. doi: 10.1016/j.mtbio.2023.100569. eCollection 2023 Apr. Mater Today Bio. 2023. PMID: 36846309 Free PMC article.
-
Role of Osteogenic Growth Peptide (OGP) and OGP(10-14) in Bone Regeneration: A Review.Int J Mol Sci. 2016 Nov 22;17(11):1885. doi: 10.3390/ijms17111885. Int J Mol Sci. 2016. PMID: 27879684 Free PMC article. Review.
-
Osteogenic growth peptide: from concept to drug design.Biopolymers. 2002;66(1):33-48. doi: 10.1002/bip.10202. Biopolymers. 2002. PMID: 12228919 Review.
Cited by
-
Mutanobactin-D, a Streptococcus mutans Non-Ribosomal Cyclic Lipopeptide, Induces Osteogenic/Odontogenic Differentiation of Human Dental Pulp Stem Cells and Human Bone Marrow Stem Cells.Int J Mol Sci. 2025 Jan 28;26(3):1144. doi: 10.3390/ijms26031144. Int J Mol Sci. 2025. PMID: 39940912 Free PMC article.
-
Advancements in GelMA bioactive hydrogels: Strategies for infection control and bone tissue regeneration.Theranostics. 2025 Jan 1;15(2):460-493. doi: 10.7150/thno.103725. eCollection 2025. Theranostics. 2025. PMID: 39744697 Free PMC article. Review.
-
Recent Advances in Bone Tissue Engineering: Enhancing the Potential of Mesenchymal Stem Cells for Regenerative Therapies.Curr Issues Mol Biol. 2025 Apr 17;47(4):287. doi: 10.3390/cimb47040287. Curr Issues Mol Biol. 2025. PMID: 40699686 Free PMC article. Review.
-
Multi-site enhancement of osteogenesis: peptide-functionalized GelMA hydrogels with three-dimensional cultures of human dental pulp stem cells.Regen Biomater. 2024 Aug 10;11:rbae090. doi: 10.1093/rb/rbae090. eCollection 2024. Regen Biomater. 2024. PMID: 39193556 Free PMC article.
-
Influence of Electrostatic Interactions on the Self-Assembly of Charged Peptides.Gels. 2025 Jan 20;11(1):80. doi: 10.3390/gels11010080. Gels. 2025. PMID: 39852051 Free PMC article.
References
-
- Bab I. A. (1995). Regulatory role of osteogenic growth peptide in proliferation, osteogenesis, and hemopoiesis. Clin. Orthop. Relat. Res. 313, 64–68. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

