Mitochondrial morphology governs ATP production rate
- PMID: 37615622
- PMCID: PMC10450615
- DOI: 10.1085/jgp.202213263
Mitochondrial morphology governs ATP production rate
Abstract
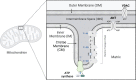
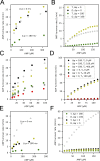
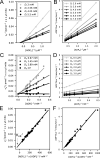
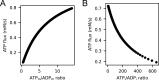
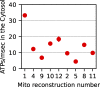
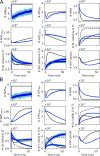
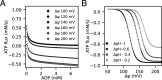
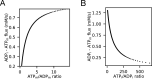
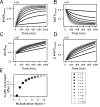
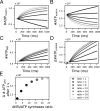
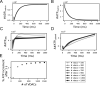
Life is based on energy conversion. In particular, in the nervous system, significant amounts of energy are needed to maintain synaptic transmission and homeostasis. To a large extent, neurons depend on oxidative phosphorylation in mitochondria to meet their high energy demand. For a comprehensive understanding of the metabolic demands in neuronal signaling, accurate models of ATP production in mitochondria are required. Here, we present a thermodynamically consistent model of ATP production in mitochondria based on previous work. The significant improvement of the model is that the reaction rate constants are set such that detailed balance is satisfied. Moreover, using thermodynamic considerations, the dependence of the reaction rate constants on membrane potential, pH, and substrate concentrations are explicitly provided. These constraints assure that the model is physically plausible. Furthermore, we explore different parameter regimes to understand in which conditions ATP production or its export are the limiting steps in making ATP available in the cytosol. The outcomes reveal that, under the conditions used in our simulations, ATP production is the limiting step and not its export. Finally, we performed spatial simulations with nine 3-D realistic mitochondrial reconstructions and linked the ATP production rate in the cytosol with morphological features of the organelles.
© 2023 Garcia et al.
Figures


Similar articles
-
Mitochondrial phosphagen kinases support the volatile power demands of motor nerve terminals.J Physiol. 2023 Dec;601(24):5705-5732. doi: 10.1113/JP284872. Epub 2023 Nov 9. J Physiol. 2023. PMID: 37942946 Free PMC article.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Neuroglobin: A promising candidate to treat neurological diseases.Neural Regen Res. 2026 Apr 1;21(4):1292-1303. doi: 10.4103/NRR.NRR-D-24-01503. Epub 2025 Jun 19. Neural Regen Res. 2026. PMID: 40536996
-
Gender differences in the context of interventions for improving health literacy in migrants: a qualitative evidence synthesis.Cochrane Database Syst Rev. 2024 Dec 12;12(12):CD013302. doi: 10.1002/14651858.CD013302.pub2. Cochrane Database Syst Rev. 2024. PMID: 39665382
Cited by
-
The Warburg Effect Explained: Integration of Enhanced Glycolysis with Heterogeneous Mitochondria to Promote Cancer Cell Proliferation.Int J Mol Sci. 2023 Oct 31;24(21):15787. doi: 10.3390/ijms242115787. Int J Mol Sci. 2023. PMID: 37958775 Free PMC article. Review.
-
Alzheimer's disease is treatable by increased cerebral blood flow (CBF) from omentum to compensate for a decreased CBF in aging.Surg Neurol Int. 2024 Oct 25;15:382. doi: 10.25259/SNI_680_2024. eCollection 2024. Surg Neurol Int. 2024. PMID: 39524577 Free PMC article. No abstract available.
-
How the Topology of the Mitochondrial Inner Membrane Modulates ATP Production.Cells. 2025 Feb 11;14(4):257. doi: 10.3390/cells14040257. Cells. 2025. PMID: 39996730 Free PMC article.
-
Spatial modeling algorithms for reactions and transport in biological cells.Nat Comput Sci. 2025 Jan;5(1):76-89. doi: 10.1038/s43588-024-00745-x. Epub 2024 Dec 19. Nat Comput Sci. 2025. PMID: 39702839 Free PMC article.
-
Origin and evolution of mitochondrial inner membrane composition.J Cell Sci. 2025 May 1;138(9):jcs263780. doi: 10.1242/jcs.263780. Epub 2025 Apr 23. J Cell Sci. 2025. PMID: 40265338 Free PMC article. Review.
References
-
- Alberts, B., Roberts K., Lewis J., Bray D., Hopkin K., Johnson A.D., Walter P., and Raff M.. 2015. Essential Cell Biology. Fourth edition. Garland Science, New York City.
-
- Balbir, T. 2007. A model of mitochondrial calcium induced calcium release. PhD thesis. Doctoral dissertation The Ohio State University, Columbus, OH. 134 pp.

