Persistent extrasynaptic hyperdopaminergia in the mouse hippocampus induces plasticity and recognition memory deficits reversed by the atypical antipsychotic sulpiride
- PMID: 37624765
- PMCID: PMC10456148
- DOI: 10.1371/journal.pone.0289770
Persistent extrasynaptic hyperdopaminergia in the mouse hippocampus induces plasticity and recognition memory deficits reversed by the atypical antipsychotic sulpiride
Abstract
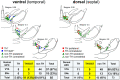
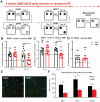
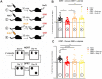
Evidence suggests that subcortical hyperdopaminergia alters cognitive function in schizophrenia and antipsychotic drugs (APD) fail at rescuing cognitive deficits in patients. In a previous study, we showed that blocking D2 dopamine receptors (D2R), a core action of APD, led to profound reshaping of mesohippocampal fibers, deficits in synaptic transmission and impairments in learning and memory in the mouse hippocampus (HP). However, it is currently unknown how excessive dopamine affects HP-related cognitive functions, and how APD would impact HP functions in such a state. After verifying the presence of DAT-positive neuronal projections in the ventral (temporal), but not in the dorsal (septal), part of the HP, GBR12935, a blocker of dopamine transporter (DAT), was infused in the CA1 of adult C57Bl/6 mice to produce local hyperdopaminergia. Chronic GBR12935 infusion in temporal CA1 induced a mild learning impairment in the Morris Water Maze and abolished long-term recognition memory in novel-object (NORT) and object-place recognition tasks (OPRT). Deficits were accompanied by a significant decrease in DAT+ mesohippocampal fibers. Intrahippocampal or systemic treatment with sulpiride during GBR infusions improved the NORT deficit but not that of OPRT. In vitro application of GBR on hippocampal slices abolished long-term depression (LTD) of fEPSP in temporal CA1. LTD was rescued by co-application with sulpiride. In conclusion, chronic DAT blockade in temporal CA1 profoundly altered mesohippocampal modulation of hippocampal functions. Contrary to previous observations in normodopaminergic mice, antagonising D2Rs was beneficial for cognitive functions in the context of hippocampal hyperdopaminergia.
Copyright: © 2023 Rocchetti et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Presynaptic D2 dopamine receptors control long-term depression expression and memory processes in the temporal hippocampus.Biol Psychiatry. 2015 Mar 15;77(6):513-25. doi: 10.1016/j.biopsych.2014.03.013. Epub 2014 Mar 20. Biol Psychiatry. 2015. PMID: 24742619
-
Parallel loss of hippocampal LTD and cognitive flexibility in a genetic model of hyperdopaminergia.Neuropsychopharmacology. 2007 Oct;32(10):2108-16. doi: 10.1038/sj.npp.1301354. Epub 2007 Mar 7. Neuropsychopharmacology. 2007. PMID: 17342172 Free PMC article.
-
Blockade of dopamine D2 receptors disrupts intrahippocampal connectivity and enhances pain-related working memory deficits in neuropathic pain rats.Eur J Pain. 2018 May;22(5):1002-1015. doi: 10.1002/ejp.1186. Epub 2018 Jan 26. Eur J Pain. 2018. PMID: 29377353
-
Nicotinic-antipsychotic drug interactions and cognitive function.EXS. 2006;98:185-205. doi: 10.1007/978-3-7643-7772-4_10. EXS. 2006. PMID: 17019889 Review.
-
[Cognition, schizophrenia and the effect of antipsychotics].Encephale. 2006 May-Jun;32(3 Pt 1):341-50. doi: 10.1016/s0013-7006(06)76162-0. Encephale. 2006. PMID: 16840928 Review. French.
Cited by
-
Comparing the prognostic value of the old and new sarcopenia criteria from the Asian Working Group on Sarcopenia in older adults with type 2 diabetes: Which set is more appropriate?Aging Clin Exp Res. 2023 Sep;35(9):1917-1926. doi: 10.1007/s40520-023-02473-0. Epub 2023 Jun 22. Aging Clin Exp Res. 2023. PMID: 37344720
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

