Chronic remote ischemic conditioning treatment in patients with chronic stable angina (EARLY-MYO-CSA): a randomized, controlled proof-of-concept trial
- PMID: 37626410
- PMCID: PMC10463998
- DOI: 10.1186/s12916-023-03041-z
Chronic remote ischemic conditioning treatment in patients with chronic stable angina (EARLY-MYO-CSA): a randomized, controlled proof-of-concept trial
Abstract
Background: Chronic remote ischemic conditioning (CRIC) has been shown to improve myocardial ischemia in experimental animal studies; however, its effectiveness in patients with chronic stable angina (CSA) has not been investigated. We conducted a proof-of-concept study to investigate the efficacy and safety of a six-month CRIC treatment in patients with CSA.
Methods: The EARLY-MYO-CSA trial was a prospective, randomized, controlled trial evaluating the CRIC treatment in patients with CSA with persistent angina pectoris despite receiving ≥ 3-month guideline-recommended optimal medical therapy. The CRIC and control groups received CRIC (at 200 mmHg) or sham CRIC (at 60 mmHg) intervention for 6 months, respectively. The primary endpoint was the 6-month change of myocardial flow reserve (MFR) on single-photon emission computed tomography. The secondary endpoints were changes in rest and stress myocardial blood flow (MBF), angina severity according to the Canadian Cardiovascular Society (CCS) classification, the Seattle Angina Questionnaire (SAQ), and a 6-min walk test (6-MWT).
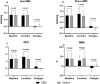
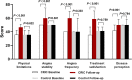
Results: Among 220 randomized CSA patients, 208 (105 in the CRIC group, and 103 in the control group) completed the treatment and endpoint assessments. The mean change in MFR was significantly greater in the CRIC group than in the control group (0.27 ± 0.38 vs. - 0.04 ± 0.25; P < 0.001). MFR increased from 1.33 ± 0.48 at baseline to 1.61 ± 0.53 (P < 0.001) in the CRIC group; however, a similar increase was not seen in the control group (1.35 ± 0.45 at baseline and 1.31 ± 0.44 at follow-up, P = 0.757). CRIC treatment, when compared with controls, demonstrated improvements in angina symptoms assessed by CCS classification (60.0% vs. 14.6%, P < 0.001), all SAQ dimensions scores (P < 0.001), and 6-MWT distances (440 [400-523] vs. 420 [330-475] m, P = 0.016). The incidence of major adverse cardiovascular events was similar between the groups.
Conclusions: CSA patients benefit from 6-month CRIC treatment with improvements in MFR, angina symptoms, and exercise performance. This treatment is well-tolerated and can be recommended for symptom relief in this clinical population.
Trial registration: [chictr.org.cn], identifier [ChiCTR2000038649].
Keywords: Chronic remote ischemic conditioning; Chronic stable angina; Coronary heart disease; Myocardial flow reserve.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
A phase II, sham-controlled, double-blinded study testing the safety and efficacy of the coronary sinus reducer in patients with refractory angina: study protocol for a randomized controlled trial.Trials. 2013 Feb 15;14:46. doi: 10.1186/1745-6215-14-46. Trials. 2013. PMID: 23413981 Free PMC article. Clinical Trial.
-
Efficacy of cardiac shock wave therapy in patients with stable angina: The design of randomized, triple blind, sham-procedure controlled study.Anatol J Cardiol. 2018 Feb;19(2):100-109. doi: 10.14744/AnatolJCardiol.2017.8023. Anatol J Cardiol. 2018. PMID: 29424731 Free PMC article. Clinical Trial.
-
A randomized, triple-blind trial of cardiac shock-wave therapy on exercise tolerance and symptoms in patients with stable angina pectoris.Coron Artery Dis. 2018 Nov;29(7):579-586. doi: 10.1097/MCA.0000000000000648. Coron Artery Dis. 2018. PMID: 29912782 Clinical Trial.
-
Efficacy and safety of Xinnaoning capsule in treating chronic stable angina (qi stagnation and blood stasis syndrome): Study protocol for a multicenter, randomized, double-blind, placebo-controlled trial.Medicine (Baltimore). 2019 Aug;98(31):e16539. doi: 10.1097/MD.0000000000016539. Medicine (Baltimore). 2019. PMID: 31374015 Free PMC article. Clinical Trial.
-
Intramyocardial bone marrow cell injection for chronic myocardial ischemia: a randomized controlled trial.JAMA. 2009 May 20;301(19):1997-2004. doi: 10.1001/jama.2009.685. JAMA. 2009. PMID: 19454638 Clinical Trial.
Cited by
-
Effect of RICAS (Remote Ischemic Preconditioning on Collaterals of Atherosclerosis Stroke): Rationale and Design.J Am Heart Assoc. 2025 Mar 18;14(6):e038570. doi: 10.1161/JAHA.124.038570. Epub 2025 Feb 19. J Am Heart Assoc. 2025. PMID: 39968799 Free PMC article.
-
RIPC improves myocardial injury by promoting mitochondrial protection via the PGC-1α/Nrf2 pathway.J Biol Chem. 2025 Jun;301(6):108476. doi: 10.1016/j.jbc.2025.108476. Epub 2025 Apr 4. J Biol Chem. 2025. PMID: 40188946 Free PMC article.
References
-
- Zhao Z, Shi Q, Guo Q, Peng L, Li X, Rao L, et al. Remote ischemic preconditioning can extend the tolerance to extended drug-coated balloon inflation time by reducing myocardial damage during percutaneous coronary intervention. Int J Cardiol. 2022;353:3–8. doi: 10.1016/j.ijcard.2022.01.049. - DOI - PubMed
-
- Heusch G, Gersh BJ. The pathophysiology of acute myocardial infarction and strategies of protection beyond reperfusion: a continual challenge. Eur Heart J. 2017;38(11):774–784. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

