Lacking Immunotherapy Biomarkers for Biliary Tract Cancer: A Comprehensive Systematic Literature Review and Meta-Analysis
- PMID: 37626908
- PMCID: PMC10453268
- DOI: 10.3390/cells12162098
Lacking Immunotherapy Biomarkers for Biliary Tract Cancer: A Comprehensive Systematic Literature Review and Meta-Analysis
Abstract
Background: Immunotherapy has recently been incorporated into the spectrum of biliary tract cancer (BTC) treatment. The identification of predictive response biomarkers is essential in order to identify those patients who may benefit most from this novel treatment option. Here, we propose a systematic literature review and a meta-analysis of PD-1, PD-L1, and other immune-related biomarker expression levels in patients with BTC.
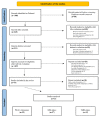
Methods: Prisma guidelines were followed for this systematic review and meta-analysis. Eligible studies were searched on PubMed. Studies published between 2017 and 2022, reporting data on PD-1/PD-L1 expression and other immune-related biomarkers in patients with BTC, were considered eligible.
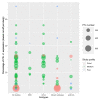
Results: A total of 61 eligible studies were identified. Despite the great heterogeneity between 39 studies reporting data on PD-L1 expression, we found a mean PD-L1 expression percentage (by choosing the lowest cut-off per study) of 25.6% (95% CI 21.0 to 30.3) in BTCs. The mean expression percentages of PD-L1 were 27.3%, 21.3%, and 27.4% in intrahepatic cholangiocarcinomas (iCCAs-15 studies), perihilar-distal CCAs (p/dCCAs-7 studies), and gallbladder cancer (GBC-5 studies), respectively. Furthermore, 4.6% (95% CI 2.38 to 6.97) and 2.5% (95% CI 1.75 to 3.34) of BTCs could be classified as TMB-H and MSI/MMRd tumors, respectively.
Conclusion: From our analysis, PD-L1 expression was found to occur approximately in 26% of BTC patients, with minimal differences based on anatomical location. TMB-H and MSI molecular phenotypes occurred less frequently. We still lack a reliable biomarker, especially in patients with mismatch-proficient tumors, and we must need to make an effort to conceive new prospective biomarker discovery studies.
Keywords: biliary cancer; immunotherapy; predictive biomarkers.
Conflict of interest statement
Angela Lamarca reports travel and educational support from Ipsen, Pfizer, Bayer, AAA, SirtEx, Novartis, Mylan, Delcath Advanz Pharma and Roche; speaker honoraria from Merck, Pfizer, Ipsen, Incyte, AAA/Novartis, QED, Servier, Astra Zeneca, EISAI, Roche, Advanz Pharma, and MSD; advisory and consultancy honoraria from EISAI, Nutricia, Ipsen, QED, Roche, Servier, Boston Scientific, Albireo Pharma, AstraZeneca, Boehringer Ingelheim, GENFIT, TransThera Biosciences, Taiho, and MSD; principal investigator-associated institutional funding from QED, Merck, Boehringer Ingelheim, Servier, Astra Zeneca, GenFit, Albireo Pharma, and Roche. She is also a member of the Knowledge Network and NETConnect Initiatives, funded by Ipsen. JMB declares research grants (from Incyte, Albireo, and Cymabay), personal lecturer fees (from Incyte and Astra Zeneca), and consultancy funding (from Albireo Pharma, Cymabay, Incyte, and OWL Metabolomics). Chiara Braconi reports speaker honoraria from Astrazeneca and Incyte; consultancy honoraria from Incyte, AstraZeneca, Serveir, and Boehringer Ingelheim; and research funds from Avacta, Medannex, and Servier. She is also a spouse to an AstraZeneca employee.
Figures


References
-
- Altekruse S.F., Petrick J.L., Rolin A.I., Cuccinelli J.E., Zou Z., Tatalovich Z., McGlynn K.A. Geographic Variation of Intrahepatic Cholangiocarcinoma, Extrahepatic Cholangiocarcinoma, and Hepatocellular Carcinoma in the United States. PLoS ONE. 2015;10:e0120574. doi: 10.1371/journal.pone.0120574. - DOI - PMC - PubMed
-
- Center for Drug Evaluation and Research. FDA D.I.S.C.O . Burst Edition: FDA Approval of Imfinzi (Durvalumab) for Adult Patients with Locally Advanced or Metastatic Biliary Tract Cancer. FDA; Silver Spring, MD, USA: 2022.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

