Efficacy and Safety of PD-1/PD-L1 Inhibitor as Single-Agent Immunotherapy in Endometrial Cancer: A Systematic Review and Meta-Analysis
- PMID: 37627060
- PMCID: PMC10452317
- DOI: 10.3390/cancers15164032
Efficacy and Safety of PD-1/PD-L1 Inhibitor as Single-Agent Immunotherapy in Endometrial Cancer: A Systematic Review and Meta-Analysis
Abstract
The programmed cell death protein 1 (PD-1)/programmed cell death ligand 1 (PD-L1) pathway plays a crucial role in the immune escape mechanism and growth of cancer cells in endometrial cancer (EC). Clinical trials investigating PD-1/PD-L1 inhibitor have shown promising results in other cancers, but their efficacy in EC still remains uncertain. Therefore, this meta-analysis aims to provide an updated and robust analysis of the effectiveness and safety of PD-1/PDL1 inhibitor as single-agent immunotherapy in EC, focusing on the objective response rate (ORR), disease control rate (DCR), and adverse events (AEs). This meta-analysis utilized STATA version 17 and RevMan version 5.4 software to pool the results of relevant studies. Five studies conducted between 2017 and 2022, comprising a total of 480 EC patients enrolled for PD-1/PD-L1 inhibitor immunotherapy met the inclusion criteria. The pooled proportion of EC patients who achieved ORR through PD-1/PD-L1 inhibitor treatment was 26.0% (95% CI: 16.0-36.0%; p < 0.05). Subgroup analysis based on mismatch repair (MMR) status showed an ORR of 44.0% (95% CI: 38.0-50.0%; p = 0.32) for the deficient mismatch repair (dMMR) group and 8.0% (95% CI: 0.0-16.0%; p = 0.07) for the proficient mismatch repair (pMMR) group. Pooled proportion analysis by DCR demonstrated an odds ratio (OR) of 41.0% (95% CI: 36.0-46.0%, p = 0.83) for patients undergoing PD-1/PD-L1 inhibitor treatment. Subgroup analysis based on MMR status revealed DCR of 54.0% (95% CI: 47.0-62.0%; p = 0.83) for the dMMR group, and 31.0% (95% CI: 25.0-39.0%; p = 0.14) for the pMMR group. The efficacy of PD-1/PD-L1 inhibitors was significantly higher in the dMMR group compared to the pMMR group, in terms of both ORR (OR = 6.30; 95% CI = 3.60-11.03; p < 0.05) and DCR (OR = 2.57; 95% CI = 1.66-3.99; p < 0.05). In terms of safety issues, the pooled proportion of patients experiencing at least one adverse event was 69.0% (95% CI: 65.0-73.0%; p > 0.05), with grade three or higher AEs occurring in 16.0% of cases (95% CI: 12.0-19.0%; p > 0.05). Based on the subgroup analysis of MMR status, PD-1/PD-L1 inhibitor immunotherapy showed significantly better efficacy among dMMR patients. These findings suggest that patients with dMMR status may be more suitable for this treatment approach. However, further research on PD-1/PD-L1 inhibitor immunotherapy strategies is needed to fully explore their potential and improve treatment outcomes in EC.
Keywords: adverse events; clinical trials efficacy; endometrial cancer; immune checkpoint inhibitor; mismatch repair; programmed cell death ligand 1; programmed cell death ligand 2; programmed cell death protein 1.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
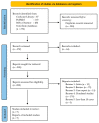
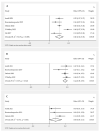
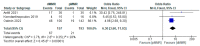


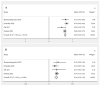

References
-
- Uterine Cancer—Cancer Stat Facts Cancer Stat Facts 2020. [(accessed on 23 May 2023)]; Available online: https://seer.cancer.gov/statfacts/html/corp.html.
-
- Kalampokas E., Giannis G., Kalampokas T., Papathanasiou A.A., Mitsopoulou D., Tsironi E., Triantafyllidou O., Gurumurthy M., Parkin D.E., Cairns M., et al. Current Approaches to the Management of Patients with Endometrial Cancer. Cancers. 2022;14:4500. doi: 10.3390/cancers14184500. - DOI - PMC - PubMed
-
- Morrison J., Balega J., Buckley L., Clamp A., Crosbie E., Drew Y., Durrant L., Forrest J., Fotopoulou C., Gajjar K., et al. British Gynaecological Cancer Society (BGCS) uterine cancer guidelines: Recommendations for practice. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022;270:50–89. doi: 10.1016/j.ejogrb.2021.11.423. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

