Proteomic Analysis on Sequential Samples of Cystic Fluid Obtained from Human Brain Tumors
- PMID: 37627098
- PMCID: PMC10452907
- DOI: 10.3390/cancers15164070
Proteomic Analysis on Sequential Samples of Cystic Fluid Obtained from Human Brain Tumors
Abstract
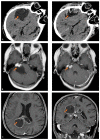
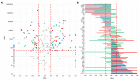
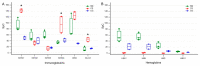
Cystic formation in human primary brain tumors is a relatively rare event whose incidence varies widely according to the histotype of the tumor. Composition of the cystic fluid has mostly been characterized in samples collected at the time of tumor resection and no indications of the evolution of cystic content are available. We characterized the evolution of the proteome of cystic fluid using a bottom-up proteomic approach on sequential samples obtained from secretory meningioma (SM), cystic schwannoma (CS) and cystic high-grade glioma (CG). We identified 1008 different proteins; 74 of these proteins were found at least once in the cystic fluid of all tumors. The most abundant proteins common to all tumors studied derived from plasma, with the exception of prostaglandin D2 synthase, which is a marker of cerebrospinal fluid origin. Overall, the protein composition of cystic fluid obtained at different times from the same tumor remained stable. After the identification of differentially expressed proteins (DEPs) and the protein-protein interaction network analysis, we identified the presence of tumor-specific pathways that may help to characterize tumor-host interactions. Our results suggest that plasma proteins leaking from local blood-brain barrier disruption are important contributors to cyst fluid formation, but cerebrospinal fluid (CSF) and the tumor itself also contribute to the cystic fluid proteome and, in some cases, as with immunoglobulin G, shows tumor-specific variations that cannot be simply explained by differences in vessel permeability or blood contamination.
Keywords: cystic fluid; cystic schwannoma (CS); glioblastoma (CG); proteomics; secretory meningioma (SM); tumor cyst.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Curtin L., Whitmire P., Rickertsen C.R., Mazza G.L., Canoll P., Johnston S.K., Mrugala M.M., Swanson K.R., Hu L.S. Assessment of Prognostic Value of Cystic Features in Glioblastoma Relative to Sex and Treatment With Standard-of-Care. Front. Oncol. 2020;10:580750. doi: 10.3389/fonc.2020.580750. - DOI - PMC - PubMed
Grants and funding
- codice ric. 08016019/Policlinico San Matteo Fondazione
- GJC2021 (n. 2022-0578)/Fondazione Cariplo
- PON ELIXIR CNRBiOMICS (PIR01_00017)/Ministry of Education, Universities and Research
- Elixir Implementation Study Proteomics 2021-23, ELIXIRxNextGenIT [PNRR, Prot. IR0000010]/Ministero Università e Ricerca (MUR)
- (RF2019-12370396)/Ministero della Salute
LinkOut - more resources
Full Text Sources

