Imaging Correlates between Headache and Breast Cancer: An [18F]FDG PET Study
- PMID: 37627174
- PMCID: PMC10453040
- DOI: 10.3390/cancers15164147
Imaging Correlates between Headache and Breast Cancer: An [18F]FDG PET Study
Abstract
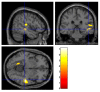
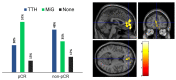
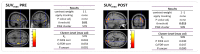
This study aimed to examine brain metabolic patterns on [18F]Fluorodeoxyglucose ([18F]FDG) positron emission tomography (PET) in breast cancer (BC), comparing patients with tension-type headache (TTH), migraine (MiG), and those without headache. Further association with BC response to neoadjuvant chemotherapy (NAC) was explored. In this prospective study, BC patients eligible for NAC performed total-body [18F]FDG PET/CT with a dedicated brain scan. A voxel-wise analysis (two-sample t-test) and a multiple regression model were used to compare brain metabolic patterns among TTH, MiG, and no-headache patients and to correlate them with clinical covariates. A single-subject analysis compared each patient's brain uptake before and after NAC with a healthy control group. Primary headache was diagnosed in 39/46 of BC patients (39% TTH and 46% MiG). TTH patients exhibited hypometabolism in specific brain regions before NAC. TTH patients with a pathological complete response (pCR) to NAC showed hypermetabolic brain regions in the anterior medial frontal cortex. The correlation between tumor uptake and brain metabolism varied before and after NAC, suggesting an inverse relationship. Additionally, the single-subject analysis revealed that hypometabolic brain regions were not present after NAC. Primary headache, especially MiG, was associated with a better response to NAC. These findings suggest complex interactions between BC, headache, and hormonal status, warranting further investigation in larger prospective cohorts.
Keywords: [18F]FDG PET/CT; brain metabolism; breast cancer; hormonal status; neoadjuvant chemotherapy; primary headache; treatment response.
Conflict of interest statement
R.D.S.: honoraria and advisory board consultancy from Lilly, Novartis, Istituto Clinico Gentili, Amgen, and Eisai (all outside the submitted work). A.S.: honoraria and advisory board consultancy from BMS (Bristol-Myers Squibb), Servier, Gilead, Pfizer, Eisai, Bayer, MSD (Merck Sharp & Dohme), Sanofi, and Incyte; speaker honoraria from Takeda, BMS, Roche, Abb-Vie, Amgen, Celgene, Servier, Gilead, AstraZeneca, Pfizer, Arqule, Lilly, Sandoz, Eisai, Novartis, Bayer, and MSD (all outside the submitted work). A.C.: personal fees from AAA, Blue Earth Diagnostics, and General Electric Healthcare (all outside the submitted work). S.M.: financial support for advisory boards and/or speaker fees from Ge Healthcare, Eli-Lilly, Novartis, and Life molecular imaging (all outside the submitted work). The other authors do not report any conflict of interest. The funders had no role in the study design; data collection and analysis; decision to publish; or preparation of the paper.
Figures

References
-
- Vos T., Flaxman A.D., Naghavi M., Lozano R., Michaud C., Ezzati M., Shibuya K., Salomon J.A., Abdalla S., Aboyans V., et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2163–2196. doi: 10.1016/S0140-6736(12)61729-2. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

