NAC Pre-Administration Prevents Cardiac Mitochondrial Bioenergetics, Dynamics, Biogenesis, and Redox Alteration in Folic Acid-AKI-Induced Cardio-Renal Syndrome Type 3
- PMID: 37627587
- PMCID: PMC10451243
- DOI: 10.3390/antiox12081592
NAC Pre-Administration Prevents Cardiac Mitochondrial Bioenergetics, Dynamics, Biogenesis, and Redox Alteration in Folic Acid-AKI-Induced Cardio-Renal Syndrome Type 3
Abstract
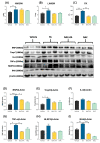
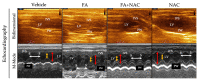
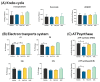
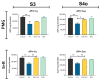
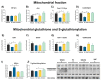
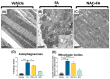
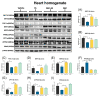
The incidence of kidney disease is increasing worldwide. Acute kidney injury (AKI) can strongly favor cardio-renal syndrome (CRS) type 3 development. However, the mechanism involved in CRS development is not entirely understood. In this sense, mitochondrial impairment in both organs has become a central axis in CRS physiopathology. This study aimed to elucidate the molecular mechanisms associated with cardiac mitochondrial impairment and its role in CRS development in the folic acid-induced AKI (FA-AKI) model. Our results showed that 48 h after FA-AKI, the administration of N-acetyl-cysteine (NAC), a mitochondrial glutathione regulator, prevented the early increase in inflammatory and cell death markers and oxidative stress in the heart. This was associated with the ability of NAC to protect heart mitochondrial bioenergetics, principally oxidative phosphorylation (OXPHOS) and membrane potential, through complex I activity and the preservation of glutathione balance, thus preventing mitochondrial dynamics shifting to fission and the decreases in mitochondrial biogenesis and mass. Our data show, for the first time, that mitochondrial bioenergetics impairment plays a critical role in the mechanism that leads to heart damage. Furthermore, NAC heart mitochondrial preservation during an AKI event can be a valuable strategy to prevent CRS type 3 development.
Keywords: NAC and mitochondria; biogenesis; cardio-renal syndrome type 3; folic acid-induced cardio-renal damage; mitochondrial ROS production; mitochondrial dynamic.
Conflict of interest statement
The authors report no conflict of interest.
Figures






Similar articles
-
Protective effects of N-acetyl-cysteine in mitochondria bioenergetics, oxidative stress, dynamics and S-glutathionylation alterations in acute kidney damage induced by folic acid.Free Radic Biol Med. 2019 Jan;130:379-396. doi: 10.1016/j.freeradbiomed.2018.11.005. Epub 2018 Nov 12. Free Radic Biol Med. 2019. PMID: 30439416
-
Chronic impairment of mitochondrial bioenergetics and β-oxidation promotes experimental AKI-to-CKD transition induced by folic acid.Free Radic Biol Med. 2020 Jul;154:18-32. doi: 10.1016/j.freeradbiomed.2020.04.016. Epub 2020 Apr 30. Free Radic Biol Med. 2020. PMID: 32360615
-
Role of mitochondria in reno-cardiac diseases: A study of bioenergetics, biogenesis, and GSH signaling in disease transition.Redox Biol. 2024 Oct;76:103340. doi: 10.1016/j.redox.2024.103340. Epub 2024 Sep 5. Redox Biol. 2024. PMID: 39250857 Free PMC article. Review.
-
Mitochondrial biogenesis: pharmacological approaches.Curr Pharm Des. 2014;20(35):5507-9. doi: 10.2174/138161282035140911142118. Curr Pharm Des. 2014. PMID: 24606795
-
Mitochondrial Dysfunction in Cardiorenal Syndrome.Antioxid Redox Signal. 2016 Aug 1;25(4):200-7. doi: 10.1089/ars.2016.6654. Antioxid Redox Signal. 2016. PMID: 27349577 Review.
Cited by
-
The role of redox signaling in mitochondria and endoplasmic reticulum regulation in kidney diseases.Arch Toxicol. 2025 May;99(5):1865-1891. doi: 10.1007/s00204-025-04041-z. Epub 2025 Apr 11. Arch Toxicol. 2025. PMID: 40214774 Free PMC article. Review.
-
Mitochondria transfer for myelin repair.J Cereb Blood Flow Metab. 2025 Mar 13:271678X251325805. doi: 10.1177/0271678X251325805. Online ahead of print. J Cereb Blood Flow Metab. 2025. PMID: 40079508 Free PMC article. Review.
-
Enhanced therapeutic effects of hypoxia-preconditioned mesenchymal stromal cell-derived extracellular vesicles in renal ischemic injury.Stem Cell Res Ther. 2025 Feb 4;16(1):39. doi: 10.1186/s13287-025-04166-z. Stem Cell Res Ther. 2025. PMID: 39901252 Free PMC article.
-
Effect of N-Acetylcysteine in Mitochondrial Function, Redox Signaling, and Sirtuin 3 Levels in the Heart During Cardiorenal Syndrome Type 4 Development.Antioxidants (Basel). 2025 Mar 20;14(3):367. doi: 10.3390/antiox14030367. Antioxidants (Basel). 2025. PMID: 40227392 Free PMC article.
References
-
- Fogo A.B., Cohen A.H., Colvin R.B., Jennette J.C., Alpers C.E. Fundamentals of Renal Pathology. 1st ed. Springer; Berlin/Heidelberg, German: 2014.
-
- Shafi T., Coresh J. Chronic Kidney Disease: Definition, Epidemiology, Cost, and Outcomes. In: Himmelfarb J., Sayegh M.H., editors. Chronic Kidney Disease, Dialysis, and Transplantation. Elsevier Inc.; Amsterdam, The Netherlands: 2010. pp. 3–21.
Grants and funding
LinkOut - more resources
Full Text Sources

