Clinical Experience with Off-Label Intrathecal Administration of Selected Antibiotics in Adults: An Overview with Pharmacometric Considerations
- PMID: 37627711
- PMCID: PMC10451962
- DOI: 10.3390/antibiotics12081291
Clinical Experience with Off-Label Intrathecal Administration of Selected Antibiotics in Adults: An Overview with Pharmacometric Considerations
Abstract
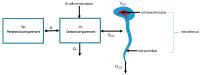
Drain-associated intracerebral infections are life-threatening emergencies. Their treatment is challenging due to the limited penetration of antibiotics to the site of infection, resulting in potentially inadequate exposure. The emergence of multidrug-resistant pathogens might force the use of off-label intrathecal (IT) doses of antibiotics. We reviewed the literature on general aspects determining intrathecal dosing regimen, using pharmacometric knowledge. We summarised clinical experience with IT doses of antibiotics that are usually not used intrathecally, as well as the outcome of the cases and concentrations reached in the cerebrospinal fluid (CSF). Factors determining the IT regimen are the size of the ventricle system and the CSF drainage volume. With regard to pharmacometrics, pharmacokinetic/pharmacodynamic indices are likely similar to those in non-cerebral infections. The following number (N) of cases were described: benzylpenicillin (>50), ampicillin (1), ceftazidime (2), cephaloridine (56), ceftriaxone (1), cefotiam (1), meropenem (57), linezolid (1), tigecycline (15), rifampicin (3), levofloxacin (2), chloramphenicol (3) and daptomycin (8). Many side effects were reported for benzylpenicillin in the 1940-50s, but for the other antibiotics, when administered correctly, all side effects were minor and reversible. These data might help when choosing an IT dosing regimen in case there is no alternative option due to antimicrobial resistance.
Keywords: antimicrobial; intracerebral infection; intrathecal administration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Efficacy of intravenous plus intrathecal/intracerebral ventricle injection of polymyxin B for post-neurosurgical intracranial infections due to MDR/XDR Acinectobacter baumannii: a retrospective cohort study.Antimicrob Resist Infect Control. 2018 Jan 19;7:8. doi: 10.1186/s13756-018-0305-5. eCollection 2018. Antimicrob Resist Infect Control. 2018. PMID: 29387342 Free PMC article.
-
Pharmacokinetics of antibacterial agents in the CSF of children and adolescents.Paediatr Drugs. 2013 Apr;15(2):93-117. doi: 10.1007/s40272-013-0017-5. Paediatr Drugs. 2013. PMID: 23529866 Review.
-
Plasma and cerebrospinal fluid population pharmacokinetic modeling and simulation of meropenem after intravenous and intrathecal administration in postoperative neurosurgical patients.Diagn Microbiol Infect Dis. 2019 Apr;93(4):386-392. doi: 10.1016/j.diagmicrobio.2018.08.003. Epub 2018 Aug 17. Diagn Microbiol Infect Dis. 2019. PMID: 30638947
-
Cefiderocol: A Siderophore Cephalosporin with Activity Against Carbapenem-Resistant and Multidrug-Resistant Gram-Negative Bacilli.Drugs. 2019 Feb;79(3):271-289. doi: 10.1007/s40265-019-1055-2. Drugs. 2019. PMID: 30712199 Review.
-
Intraventricular and lumbar intrathecal administration of antibiotics in postneurosurgical patients with meningitis and/or ventriculitis in a serious clinical state.J Neurosurg. 2013 Dec;119(6):1596-602. doi: 10.3171/2013.6.JNS122126. Epub 2013 Aug 16. J Neurosurg. 2013. PMID: 23952688
Cited by
-
New Antibiotics for the Treatment of Nosocomial Central Nervous System Infections.Antibiotics (Basel). 2024 Jan 7;13(1):58. doi: 10.3390/antibiotics13010058. Antibiotics (Basel). 2024. PMID: 38247617 Free PMC article. Review.
-
Comprehensive Therapeutic Approaches to Tuberculous Meningitis: Pharmacokinetics, Combined Dosing, and Advanced Intrathecal Therapies.Pharmaceutics. 2024 Apr 14;16(4):540. doi: 10.3390/pharmaceutics16040540. Pharmaceutics. 2024. PMID: 38675201 Free PMC article. Review.
References
-
- Neuberger A., Shofty B., Bishop B., Naffaa M.E., Binawi T., Babich T., Rappaport Z.H., Zaaroor M., Sviri G., Yahav D., et al. Risk factors associated with death or neurological deterioration among patients with Gram-negative postneurosurgical meningitis. Clin. Microbiol. Infect. 2016;22:573.e1–573.e4. doi: 10.1016/j.cmi.2016.03.016. - DOI - PubMed
-
- Zheng G., Shi Y., Cao Y., Qian L., Lv H., Zhang L., Zhang G. Clinical Feature, Therapy, Antimicrobial Resistance Gene Distribution, and Outcome of Nosocomial Meningitis Induced by Multidrug-Resistant Enterobacteriaceae-A Longitudinal Cohort Study From Two Neurosurgical Centers in Northern China. Front. Cell. Infect. Microbiol. 2022;12:839257. doi: 10.3389/fcimb.2022.839257. - DOI - PMC - PubMed
-
- de Champs C., Guelon D., Joyon D., Sirot D., Chanal M., Sirot J. Treatment of a meningitis due to an Enterobacter aerogenes producing a derepressed cephalosporinase and a Klebsiella pneumoniae producing an extended-spectrum beta-lactamase. Infection. 1991;19:181–183. doi: 10.1007/BF01643247. - DOI - PubMed
-
- Viladrich P.F., Cabellos C., Pallares R., Tubau F., Martinez-Lacasa J., Linares J., Gudiol F. High doses of cefotaxime in treatment of adult meningitis due to Streptococcus pneumoniae with decreased susceptibilities to broad-spectrum cephalosporins. Antimicrob. Agents Chemother. 1996;40:218–220. doi: 10.1128/AAC.40.1.218. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

