CD155 and Its Receptors as Targets for Cancer Therapy
- PMID: 37629138
- PMCID: PMC10455395
- DOI: 10.3390/ijms241612958
CD155 and Its Receptors as Targets for Cancer Therapy
Abstract
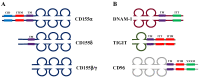
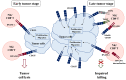
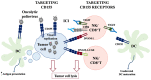
CD155, also known as the poliovirus receptor, is an adhesion molecule often overexpressed in tumors of different origins where it promotes cell migration and proliferation. In addition to this pro-tumorigenic function, CD155 plays an immunomodulatory role during tumor progression since it is a ligand for both the activating receptor DNAM-1 and the inhibitory receptor TIGIT, expressed on cytotoxic innate and adaptative lymphocytes. DNAM-1 is a well-recognized receptor involved in anti-tumor immune surveillance. However, in advanced tumor stages, TIGIT is up-regulated and acts as an immune checkpoint receptor, counterbalancing DNAM-1-mediated cancer cell clearance. Pre-clinical studies have proposed the direct targeting of CD155 on tumor cells as well as the enhancement of DNAM-1-mediated anti-tumor functions as promising therapeutic approaches. Moreover, immunotherapeutic use of anti-TIGIT blocking antibody alone or in combined therapy has already been included in clinical trials. The aim of this review is to summarize all these potential therapies, highlighting the still controversial role of CD155 during tumor progression.
Keywords: cancer therapy; poliovirus receptor; tumor immune surveillance.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Iwasaki A., Welker R., Mueller S., Linehan M., Nomoto A., Wimmer E. Immunofluorescence analysis of poliovirus receptor expression in Peyer’s patches of humans, primates, and CD155 transgenic mice: Implications for poliovirus infection. J. Infect. Dis. 2002;186:585–592. doi: 10.1086/342682. - DOI - PubMed
-
- Maier M.K., Seth S., Czeloth N., Qiu Q., Ravens I., Kremmer E., Ebel M., Müller W., Pabst O., Förster R., et al. The adhesion receptor CD155 determines the magnitude of humoral immune responses against orally ingested antigens. Eur. J. Immunol. 2007;37:2214–2225. doi: 10.1002/eji.200737072. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

