Differences in the Susceptibility of Human Tubular Epithelial Cells for Infection with Orthohantaviruses
- PMID: 37632012
- PMCID: PMC10459294
- DOI: 10.3390/v15081670
Differences in the Susceptibility of Human Tubular Epithelial Cells for Infection with Orthohantaviruses
Abstract
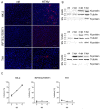
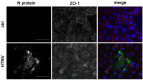
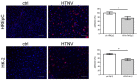
Diseases induced by infection with pathogenic orthohantaviruses are characterized by a pronounced organ-specific manifestation. Pathogenic Eurasian orthohantaviruses cause hemorrhagic fever with renal syndrome (HFRS) with often massive proteinuria. Therefore, the use of a relevant kidney cell culture would be favorable to analyze the underlying cellular mechanisms of orthohantavirus-induced acute kidney injury (AKI). We tested different human tubular epithelial cell lines for their suitability as an in vitro infection model. Permissiveness and replication kinetics of highly pathogenic Hantaan virus (HTNV) and non-/low-pathogenic Tula virus (TULV) were analyzed in tubular epithelial cell lines and compared to human primary tubular epithelial cells. Ana-lysis of the cell line HK-2 revealed the same results for viral replication, morphological and functional effects as observed for HTNV in primary cells. In contrast, the cell lines RPTEC/TERT1 and TH1 demonstrated only poor infection rates after inoculation with HTNV and are unusable as an infection model. While pathogenic HNTV infects primary tubular and HK-2 cells, non-/low-pathogenic TULV infects neither primary tubular cells nor the cell line HK-2. Our results show that permissiveness of renal cells varies between orthohantaviruses with differences in pathogenicity and that HK-2 cells demonstrate a suitable in vitro model to study viral tropism and pathogenesis of orthohantavirus-induced AKI.
Keywords: Hantaan virus; Tula virus; acute kidney injury; cell-to-cell contacts; kidney; orthohantavirus; pathogenesis; receptor; tubular epithelium.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures




Similar articles
-
Replication kinetics of pathogenic Eurasian orthohantaviruses in human mesangial cells.Virol J. 2024 Oct 1;21(1):241. doi: 10.1186/s12985-024-02517-5. Virol J. 2024. PMID: 39354507 Free PMC article.
-
Virus- and cell type-specific effects in orthohantavirus infection.Virus Res. 2019 Jan 15;260:102-113. doi: 10.1016/j.virusres.2018.11.015. Epub 2018 Nov 30. Virus Res. 2019. PMID: 30508604
-
Cells of the human respiratory tract support the replication of pathogenic Old World orthohantavirus Puumala.Virol J. 2021 Aug 17;18(1):169. doi: 10.1186/s12985-021-01636-7. Virol J. 2021. PMID: 34404450 Free PMC article.
-
Orthohantavirus Pathogenesis and Cell Tropism.Front Cell Infect Microbiol. 2020 Aug 4;10:399. doi: 10.3389/fcimb.2020.00399. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32903721 Free PMC article. Review.
-
Hemorrhagic Fever with Renal Syndrome in Asia: History, Pathogenesis, Diagnosis, Treatment, and Prevention.Viruses. 2023 Feb 18;15(2):561. doi: 10.3390/v15020561. Viruses. 2023. PMID: 36851775 Free PMC article. Review.
Cited by
-
Hantaan virus glycoprotein Gc induces NEDD4-dependent PTEN ubiquitination and degradation to escape the restriction of autophagosomes and facilitate viral propagation.FASEB J. 2025 Jan 15;39(1):e70295. doi: 10.1096/fj.202401916R. FASEB J. 2025. PMID: 39792131 Free PMC article.
-
Replication kinetics of pathogenic Eurasian orthohantaviruses in human mesangial cells.Virol J. 2024 Oct 1;21(1):241. doi: 10.1186/s12985-024-02517-5. Virol J. 2024. PMID: 39354507 Free PMC article.
References
-
- Klempa B., Avsic-Zupanc T., Clement J., Dzagurova T.K., Henttonen H., Heyman P., Jakab F., Krüger D.H., Maes P., Papa A., et al. Complex evolution and epidemiology of Dobrava-Belgrade hantavirus: Definition of genotypes and their characteristics. Arch. Virol. 2013;158:521–529. doi: 10.1007/s00705-012-1514-5. - DOI - PMC - PubMed
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources

