Remibrutinib (LOU064) inhibits neuroinflammation driven by B cells and myeloid cells in preclinical models of multiple sclerosis
- PMID: 37633912
- PMCID: PMC10463946
- DOI: 10.1186/s12974-023-02877-9
Remibrutinib (LOU064) inhibits neuroinflammation driven by B cells and myeloid cells in preclinical models of multiple sclerosis
Abstract
Background: Bruton's tyrosine kinase (BTK) is a key signaling node in B cell receptor (BCR) and Fc receptor (FcR) signaling. BTK inhibitors (BTKi) are an emerging oral treatment option for patients suffering from multiple sclerosis (MS). Remibrutinib (LOU064) is a potent, highly selective covalent BTKi with a promising preclinical and clinical profile for MS and other autoimmune or autoallergic indications.
Methods: The efficacy and mechanism of action of remibrutinib was assessed in two different experimental autoimmune encephalomyelitis (EAE) mouse models for MS. The impact of remibrutinib on B cell-driven EAE pathology was determined after immunization with human myelin oligodendrocyte glycoprotein (HuMOG). The efficacy on myeloid cell and microglia driven neuroinflammation was determined in the RatMOG EAE. In addition, we assessed the relationship of efficacy to BTK occupancy in tissue, ex vivo T cell response, as well as single cell RNA-sequencing (scRNA-seq) in brain and spinal cord tissue.
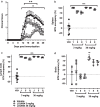
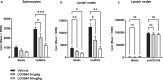
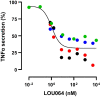
Results: Remibrutinib inhibited B cell-dependent HuMOG EAE in dose-dependent manner and strongly reduced neurological symptoms. At the efficacious oral dose of 30 mg/kg, remibrutinib showed strong BTK occupancy in the peripheral immune organs and in the brain of EAE mice. Ex vivo MOG-specific T cell recall response was reduced, but not polyclonal T cell response, indicating absence of non-specific T cell inhibition. Remibrutinib also inhibited RatMOG EAE, suggesting that myeloid cell and microglia inhibition contribute to its efficacy in EAE. Remibrutinib did not reduce B cells, total Ig levels nor MOG-specific antibody response. In brain and spinal cord tissue a clear anti-inflammatory effect in microglia was detected by scRNA-seq. Finally, remibrutinib showed potent inhibition of in vitro immune complex-driven inflammatory response in human microglia.
Conclusion: Remibrutinib inhibited EAE models by a two-pronged mechanism based on inhibition of pathogenic B cell autoreactivity, as well as direct anti-inflammatory effects in microglia. Remibrutinib showed efficacy in both models in absence of direct B cell depletion, broad T cell inhibition or reduction of total Ig levels. These findings support the view that remibrutinib may represent a novel treatment option for patients with MS.
Keywords: Autoimmunity; BTK; LOU064; Multiple sclerosis; Neuroinflammation; Remibrutinib.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
All authors were Novartis employees at the time of the studies.
Figures




References
-
- Compston A, Coles A. Multiple sclerosis. Lancet. 2008;372:1502–1517. - PubMed
-
- Kutzelnigg A, Lassmann H. Pathology of multiple sclerosis and related inflammatory demyelinating diseases. Handb Clin Neurol. 2014;122:15–58. - PubMed
-
- Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol. 2005;23:683–747. - PubMed
-
- Archelos JJ, Storch MK, Hartung HP. The role of B cells and autoantibodies in multiple sclerosis. Ann Neurol. 2000;47:694–706. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

