ALCAM on human oligodendrocytes mediates CD4 T cell adhesion
- PMID: 37640028
- PMCID: PMC10766241
- DOI: 10.1093/brain/awad286
ALCAM on human oligodendrocytes mediates CD4 T cell adhesion
Abstract
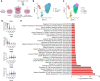
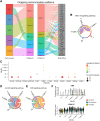
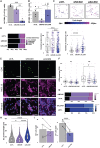
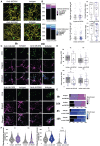
Multiple sclerosis is a chronic neuroinflammatory disorder characterized by demyelination, oligodendrocyte damage/loss and neuroaxonal injury in the context of immune cell infiltration in the CNS. No neuroprotective therapy is available to promote the survival of oligodendrocytes and protect their myelin processes in immune-mediated demyelinating diseases. Pro-inflammatory CD4 Th17 cells can interact with oligodendrocytes in multiple sclerosis and its animal model, causing injury to myelinating processes and cell death through direct contact. However, the molecular mechanisms underlying the close contact and subsequent detrimental interaction of Th17 cells with oligodendrocytes remain unclear. In this study we used single cell RNA sequencing, flow cytometry and immunofluorescence studies on CNS tissue from multiple sclerosis subjects, its animal model and controls to characterize the expression of cell adhesion molecules by mature oligodendrocytes. We found that a significant proportion of human and murine mature oligodendrocytes express melanoma cell adhesion molecule (MCAM) and activated leukocyte cell adhesion molecule (ALCAM) in multiple sclerosis, in experimental autoimmune encephalomyelitis and in controls, although their regulation differs between human and mouse. We observed that exposure to pro-inflammatory cytokines or to human activated T cells are associated with a marked downregulation of the expression of MCAM but not of ALCAM at the surface of human primary oligodendrocytes. Furthermore, we used in vitro live imaging, immunofluorescence and flow cytometry to determine the contribution of these molecules to Th17-polarized cell adhesion and cytotoxicity towards human oligodendrocytes. Silencing and blocking ALCAM but not MCAM limited prolonged interactions between human primary oligodendrocytes and Th17-polarized cells, resulting in decreased adhesion of Th17-polarized cells to oligodendrocytes and conferring significant protection of oligodendrocytic processes. In conclusion, we showed that human oligodendrocytes express MCAM and ALCAM, which are differently modulated by inflammation and T cell contact. We found that ALCAM is a ligand for Th17-polarized cells, contributing to their capacity to adhere and induce damage to human oligodendrocytes, and therefore could represent a relevant target for neuroprotection in multiple sclerosis.
Keywords: ALCAM; MCAM; Th17 cells; multiple sclerosis; oligodendrocytes.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
The authors report no competing interests.
Figures


References
-
- Kuhlmann T, Ludwin S, Prat A, Antel J, Bruck W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017;133:13–24. - PubMed
-
- Cui QL, Khan D, Rone M, et al. Sublethal oligodendrocyte injury: A reversible condition in multiple sclerosis? Ann Neurol. 2017;81:811–824. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

