Nature-inspired nanocarriers for improving drug therapy of atherosclerosis
- PMID: 37641591
- PMCID: PMC10460486
- DOI: 10.1093/rb/rbad069
Nature-inspired nanocarriers for improving drug therapy of atherosclerosis
Abstract
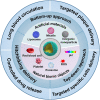
Atherosclerosis (AS) has emerged as one of the prevalent arterial vascular diseases characterized by plaque and inflammation, primarily causing disability and mortality globally. Drug therapy remains the main treatment for AS. However, a series of obstacles hinder effective drug delivery. Nature, from natural micro-/nano-structural biological particles like natural cells and extracellular vesicles to the distinctions between the normal and pathological microenvironment, offers compelling solutions for efficient drug delivery. Nature-inspired nanocarriers of synthetic stimulus-responsive materials and natural components, such as lipids, proteins and membrane structures, have emerged as promising candidates for fulfilling drug delivery needs. These nanocarriers offer several advantages, including prolonged blood circulation, targeted plaque delivery, targeted specific cells delivery and controlled drug release at the action site. In this review, we discuss the nature-inspired nanocarriers which leverage the natural properties of cells or the microenvironment to improve atherosclerotic drug therapy. Finally, we provide an overview of the challenges and opportunities of applying these innovative nature-inspired nanocarriers.
Keywords: atherosclerosis; drug delivery; membrane-coating; nature-inspired nanocarriers; stimuli-responsive.
© The Author(s) 2023. Published by Oxford University Press.
Figures







Similar articles
-
Bio-Inspired Nanocarriers Derived from Stem Cells and Their Extracellular Vesicles for Targeted Drug Delivery.Pharmaceutics. 2023 Jul 24;15(7):2011. doi: 10.3390/pharmaceutics15072011. Pharmaceutics. 2023. PMID: 37514197 Free PMC article. Review.
-
Exosome-based nanocarriers as bio-inspired and versatile vehicles for drug delivery: recent advances and challenges.J Mater Chem B. 2019 Apr 21;7(15):2421-2433. doi: 10.1039/c9tb00170k. Epub 2019 Mar 13. J Mater Chem B. 2019. PMID: 32255119 Review.
-
Unleashing the biomimetic targeting potential of platelet-derived nanocarriers on atherosclerosis.Colloids Surf B Biointerfaces. 2024 Aug;240:113979. doi: 10.1016/j.colsurfb.2024.113979. Epub 2024 May 22. Colloids Surf B Biointerfaces. 2024. PMID: 38823339 Review.
-
Stimuli-responsive and biomimetic delivery systems for sepsis and related complications.J Control Release. 2022 Dec;352:1048-1070. doi: 10.1016/j.jconrel.2022.11.013. Epub 2022 Nov 17. J Control Release. 2022. PMID: 36372385 Review.
-
Nanocarriers surface engineered with cell membranes for cancer targeted chemotherapy.J Nanobiotechnology. 2022 Jan 21;20(1):45. doi: 10.1186/s12951-022-01251-w. J Nanobiotechnology. 2022. PMID: 35062958 Free PMC article. Review.
Cited by
-
Targeting mitochondrial dynamics and redox regulation in cardiovascular diseases.Trends Pharmacol Sci. 2024 Apr;45(4):290-303. doi: 10.1016/j.tips.2024.02.001. Epub 2024 Mar 7. Trends Pharmacol Sci. 2024. PMID: 38458847 Free PMC article. Review.
-
β-Cyclodextrin-based nanoassemblies for the treatment of atherosclerosis.Regen Biomater. 2024 Jun 17;11:rbae071. doi: 10.1093/rb/rbae071. eCollection 2024. Regen Biomater. 2024. PMID: 38966400 Free PMC article. Review.
-
Cancer cell response to extrinsic and intrinsic mechanical cue: opportunities for tumor apoptosis strategies.Regen Biomater. 2024 Feb 20;11:rbae016. doi: 10.1093/rb/rbae016. eCollection 2024. Regen Biomater. 2024. PMID: 38476678 Free PMC article. Review.
-
How Advanced Is Nanomedicine for Atherosclerosis?Int J Nanomedicine. 2025 Mar 17;20:3445-3470. doi: 10.2147/IJN.S508757. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40125442 Free PMC article. Review.
-
A lipid/PLGA nanocomplex to reshape tumor immune microenvironment for colon cancer therapy.Regen Biomater. 2024 Mar 28;11:rbae036. doi: 10.1093/rb/rbae036. eCollection 2024. Regen Biomater. 2024. PMID: 38628547 Free PMC article.
References
-
- Zhang S, Liu Y, Cao Y, Zhang S, Sun J, Wang Y, Song S, Zhang H.. Targeting the microenvironment of vulnerable atherosclerotic plaques: an emerging diagnosis and therapy strategy for atherosclerosis. Adv Mater 2022;34:e2110660. - PubMed
-
- Song P, Fang Z, Wang H, Cai Y, Rahimi K, Zhu Y, Fowkes FGR, Fowkes FJI, Rudan I.. Global and regional prevalence, burden, and risk factors for carotid atherosclerosis: a systematic review, meta-analysis, and modelling study. Lancet Glob Health 2020;8:e721–9. - PubMed
-
- Libby P. The changing landscape of atherosclerosis. Nature 2021;592:524–33. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

