Safety and efficacy of topical tacrolimus 0.03% in the management of vernal keratoconjunctivitis: a non-randomized controlled clinical trial
- PMID: 37641784
- PMCID: PMC10445306
- DOI: 10.51329/mehdiophthal1446
Safety and efficacy of topical tacrolimus 0.03% in the management of vernal keratoconjunctivitis: a non-randomized controlled clinical trial
Abstract
Background: Vernal keratoconjunctivitis (VKC) is a bilateral, recurrent, chronic conjunctival inflammatory disease with seasonal exacerbations. This study aimed to assess the efficacy and safety of tacrolimus 0.03% eye ointment in the management of chronic VKC.
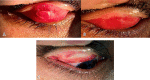
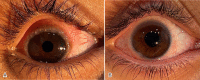
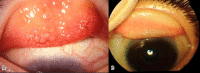
Methods: This was an open-label, prospective, non-randomized, comparative interventional study that enrolled 50 patients with chronic VKC, who were allocated to one of two groups. The first group was treated with tacrolimus 0.03% eye ointment twice daily for 2 months then once daily for 2 months, followed by once every other day for another 2 months. The control group was treated with standard anti-allergic drugs, topical fluorometholone 0.1% eye drops three times daily for 2 weeks and gradually tapered for another 2 weeks, with topical olopatadine 0.1% administered twice daily during the follow-up period. Disease severity was assessed using a four-point scale for symptoms and signs. Treatment efficacy was assessed by analyzing changes in symptoms and signs, and by clinical photography.
Results: Fifty patients with bilateral chronic VKC completed the follow-up. The mean (standard deviation) ages of the tacrolimus and control groups were comparable (16.20 [5.10] years versus 16.48 [4.19] years, P > 0.05). The most commonly reported symptom was itching, and the most common signs were papillary hypertrophy and conjunctival hyperemia. All symptoms and signs were significantly reduced after treatment in both groups. The tacrolimus group showed a more significant improvement at 3 and 6 months in the mean composite symptom score (both P < 0.05) and in the mean composite sign score (both P < 0.05). Regarding complications, one case of increased intraocular pressure occurred in the control group (4%) after 2 weeks of steroid treatment, while there were no complications in the tacrolimus group, except for some reports of stinging sensation, which was well tolerated.
Conclusions: Treatment of chronic bilateral VKC with tacrolimus 0.03% eye ointment is effective and safe. It could be considered an alternative treatment to reduce steroid-associated complications in patients with chronic VKC. Future double-blinded clinical trials with a longer follow-up period are necessary to confirm our findings and to determine the long-term safety of topical tacrolimus 0.03% ointment in VKC.
Keywords: fluorometholone; prospective study; steroid; tacrolimus anhydrous; vernal keratoconjunctivitis.
© Author(s).
Conflict of interest statement
None.
Figures



References
-
- Samyukta SK, Pawar N, Ravindran M, Allapitchai F, Rengappa R. Monotherapy of topical tacrolimus 0 03% in the treatment of vernal keratoconjunctivitis in the pediatric population. J AAPOS. 2019;23(1):36.e1–36. - PubMed
-
- Arsalan Z, Zafar O, Naqvi SAH, Ain QU. Effectiveness of steroids versus immunomodulators intreatment of vernal keratoConjunctivitis A Study at Armed Forces Institute of Ophthalmology. Pak Armed Forces Med J. 2021;71(6):2045–2048.
LinkOut - more resources
Full Text Sources
