A unifying mechanism for protein transport through the core bacterial Sec machinery
- PMID: 37643640
- PMCID: PMC10465204
- DOI: 10.1098/rsob.230166
A unifying mechanism for protein transport through the core bacterial Sec machinery
Abstract
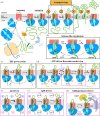
Encapsulation and compartmentalization are fundamental to the evolution of cellular life, but they also pose a challenge: how to partition the molecules that perform biological functions-the proteins-across impermeable barriers into sub-cellular organelles, and to the outside. The solution lies in the evolution of specialized machines, translocons, found in every biological membrane, which act both as gate and gatekeeper across and into membrane bilayers. Understanding how these translocons operate at the molecular level has been a long-standing ambition of cell biology, and one that is approaching its denouement; particularly in the case of the ubiquitous Sec system. In this review, we highlight the fruits of recent game-changing technical innovations in structural biology, biophysics and biochemistry to present a largely complete mechanism for the bacterial version of the core Sec machinery. We discuss the merits of our model over alternative proposals and identify the remaining open questions. The template laid out by the study of the Sec system will be of immense value for probing the many other translocons found in diverse biological membranes, towards the ultimate goal of altering or impeding their functions for pharmaceutical or biotechnological purposes.
Keywords: Sec machinery; SecA; SecYEG; bacterial secretion; protein transport.
Conflict of interest statement
We declare we have no competing interests.
Figures


Similar articles
-
Single-molecule analysis of dynamics and interactions of the SecYEG translocon.FEBS J. 2021 Apr;288(7):2203-2221. doi: 10.1111/febs.15596. Epub 2020 Oct 29. FEBS J. 2021. PMID: 33058437
-
Coassembly of SecYEG and SecA Fully Restores the Properties of the Native Translocon.J Bacteriol. 2018 Dec 7;201(1):e00493-18. doi: 10.1128/JB.00493-18. Print 2019 Jan 1. J Bacteriol. 2018. PMID: 30275279 Free PMC article.
-
The Sec translocon mediated protein transport in prokaryotes and eukaryotes.Mol Membr Biol. 2014 Mar-May;31(2-3):58-84. doi: 10.3109/09687688.2014.907455. Mol Membr Biol. 2014. PMID: 24762201 Review.
-
Dynamic interaction of the sec translocon with the chaperone PpiD.J Biol Chem. 2014 Aug 1;289(31):21706-15. doi: 10.1074/jbc.M114.577916. Epub 2014 Jun 20. J Biol Chem. 2014. PMID: 24951590 Free PMC article.
-
The way is the goal: how SecA transports proteins across the cytoplasmic membrane in bacteria.FEMS Microbiol Lett. 2018 Jun 1;365(11):fny093. doi: 10.1093/femsle/fny093. FEMS Microbiol Lett. 2018. PMID: 29790985 Free PMC article. Review.
Cited by
-
The MitoLuc assay for the analysis of the mechanism of mitochondrial protein import.Methods Enzymol. 2024;706:407-436. doi: 10.1016/bs.mie.2024.07.033. Epub 2024 Aug 22. Methods Enzymol. 2024. PMID: 39455227 Free PMC article.
-
Redundant essentiality of AsmA-like proteins in Pseudomonas aeruginosa.mSphere. 2024 Feb 28;9(2):e0067723. doi: 10.1128/msphere.00677-23. Epub 2024 Feb 2. mSphere. 2024. PMID: 38305166 Free PMC article.
-
Advancements in Escherichia coli secretion systems for enhanced recombinant protein production.World J Microbiol Biotechnol. 2025 Mar 3;41(3):90. doi: 10.1007/s11274-025-04302-0. World J Microbiol Biotechnol. 2025. PMID: 40025370 Review.
-
Whole Cell Luminescence-Based Screen for Inhibitors of the Bacterial Sec Machinery.Biochemistry. 2024 Sep 17;63(18):2344-2351. doi: 10.1021/acs.biochem.4c00264. Epub 2024 Aug 29. Biochemistry. 2024. PMID: 39207823 Free PMC article.
References
-
- Blobel G, Sabatini DD. 1971. Ribosome–membrane interaction in eukaryotic cells. In Biomembranes (ed. Manson LA), pp. 193-195. New York, NY: Springer.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

